JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Gabriel Phthalimide Synthesis is considered one of the most asked concept.
34 Questions around this concept.
The electrolytic reduction of nitrobenzene in strongly acidic medium produces:-
Which of the following is NOT a correct method of the preparation of benzylamine from cyanobenzene?
Method by which Aniline cannot be prepared is :
Re-NEET 2026: 30-Day Study Plan PDF | Last 5 Year's PYQ's with Solutions
Re-NEET 2026: NEET Previous 17 Years QP (2009–2025) | Most Scoring Concepts
Re-NEET 2026: Crash Course for Re-NEET 2026 | 100% FREE | Mock Test
The increasing order of nitration of the following compounds is :

$\mathrm{C}_2 \mathrm{H}_5-\mathrm{Cl} \xrightarrow{\mathrm{KCN}} A \xrightarrow{\mathrm{H}_3 \mathrm{O}^{+}} B \xrightarrow[(i) \Delta]{(i) \mathrm{NH}_3} C \xrightarrow[\mathrm{~N}_2 \mathrm{OH}]{\mathrm{Br}_2} D$
The product D is

Major product is :
In the Hofmann bromamide degradation reaction, the number of moles of NaOH and Br2 used per mole of amine produced are :
Acetamide is treated with the following reagents separately. Which one of these would yield methylamine?
Curtius Reaction
Acid azides on heating in non-polar solvent give alkyl isocyanate via acylnitrene formation, which on hydrolysis gives 1o amine. The reaction occurs as follows:

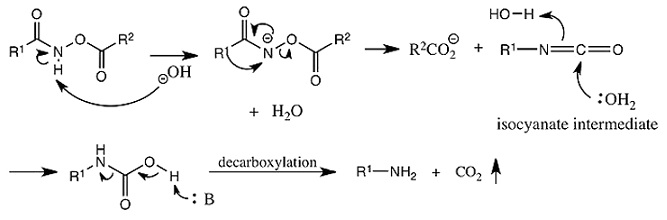
Schmidt Reaction
Carboxylic acid on reaction with hydrazoic acid in the presence of acid(H2SO4) gives acid azide which on heating gives alkyl isocyanate followed bu hydrolysis to give 1o amine. The reaction occurs as follows:
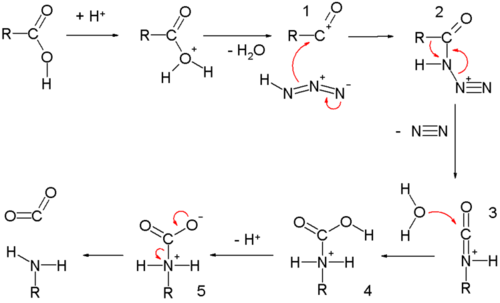
Lossen Reaction
Hydroxamic acid in basic medium rearranges to give alkyl isocyanate via acylnitrene formation, which on hydrolysis gives 1o amine. The reaction occurs as follows:
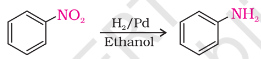
Reduction of Nitroalkanes
Nitro compounds are reduced to amines by reduction with metals(Fe, Sn or Zn) in dil. HCl or SnCl2 in HCl or by passing H2 gas in the presence of finely divided Ni, Pt, or Pd. Reduction with Fe scrap and HCl is preferred because the FeCl2 formed gets hydrolysed to give HCl during the reaction, and thus only a small amount of HCl is required for the initiation of the reaction. The reactions occur as follows:

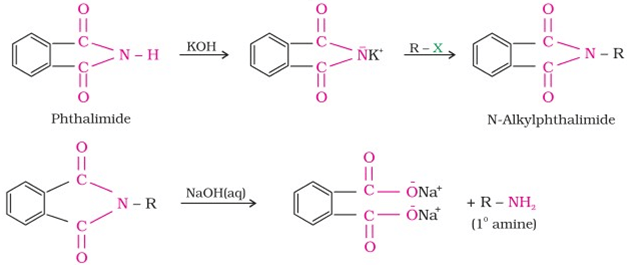
This reaction is used for the preparation of 1o aliphatic amine and 1o aromatic amine. Phthalimide on treatment with ethanolic KOH forms potassium salt of phthalimide which on heating with RX followed by either alkaline hydrolysis or hydrazinolysis with hydrazine(H2N.NH2) produces the corresponding 1o amine. 1o aromatic amine cannot be synthesised by this method because ArX does not undergo SN reaction with anion formed by phthalimide. The reaction occurs as follows:
Amides on reaction with Br2 or Cl2 in alkali give 1o amine with one C atom less than the parent amide. This is known as Hofmann bromamide rearrangement or degradation reaction. The reaction occurs as follows:
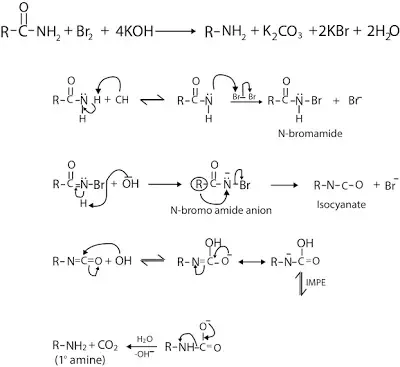
Since only primary amides undergo Hoffmann Bromamide reaction but secondary amides do not undergo this reaction. But secondary diamide undergoes this reaction. Thus, it is consider as the special case of Hoffmann Bromamide reaction. The reaction occurs as follows:

"Stay in the loop. Receive exam news, study resources, and expert advice!"