Hydroboration and Oxidation MCQ - Practice Questions with Answers
Quick Facts
-
12 Questions around this concept.
Solve by difficulty
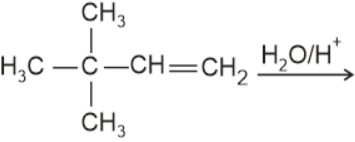
The major product is:
Concepts Covered - 2
Hydroboration-oxidation serves as an important method for synthesis of alcohol(1o & 2o). The reaction occurs as follows:

The addition of boron hydride is syn-addition. It is generally carried out by BH3 (boron hydride) B2H6 (diborane) in THF. In each addition, the boron atom becomes attached to the less substituted carbon atom of double bond and H is transferred from boron atom to the other carbon atom of double bond. Thus it follows Anti-Markonikoff’s addition.
Cold concentrated sulphuric acid adds to alkenes in accordance with Markovnikov rule to form alkyl hydrogen sulphate by the electrophilic addition reaction.
For example:
Mechanism: In this reaction, carbon-carbon double bond is broken first. Then one of the H+ is released and combines with one of the carbon. Now, a carbocation is already formed after the breaking of double bond. Now, if the carbocation has a possibility to achieve more stability, then first it becomes more stable either by hydride shift or methyl shift. Then HSO-4 binds with carbocation and forms the final product as given below.
Study it with Videos
"Stay in the loop. Receive exam news, study resources, and expert advice!"

