- Home
- Study Material
- Intramolecular and Intermolecular Bonds MCQ - Practice Questions with Answers
Quick Facts
-
7 Questions around this concept.
Solve by difficulty
Polar covalent bonds are formed due to
Concepts Covered - 1
Intramolecular and Intermolecular Bonds
Intramolecular Bonds:
In any given molecule, the atoms interact with each other via intramolecular bonds.
- 1. Covalent Bonds:
- These are the bonds that hold the atoms together in a molecule.
- These are the strong bonds that cannot be broken except for by the enzymatic actions.
- There are two types of covalent bonds:
1.1. Non-Polar Covalent Bonds:
- These bonds are formed when two atoms share electrons equally such that their electronegativity value is also equal. For example, H2:
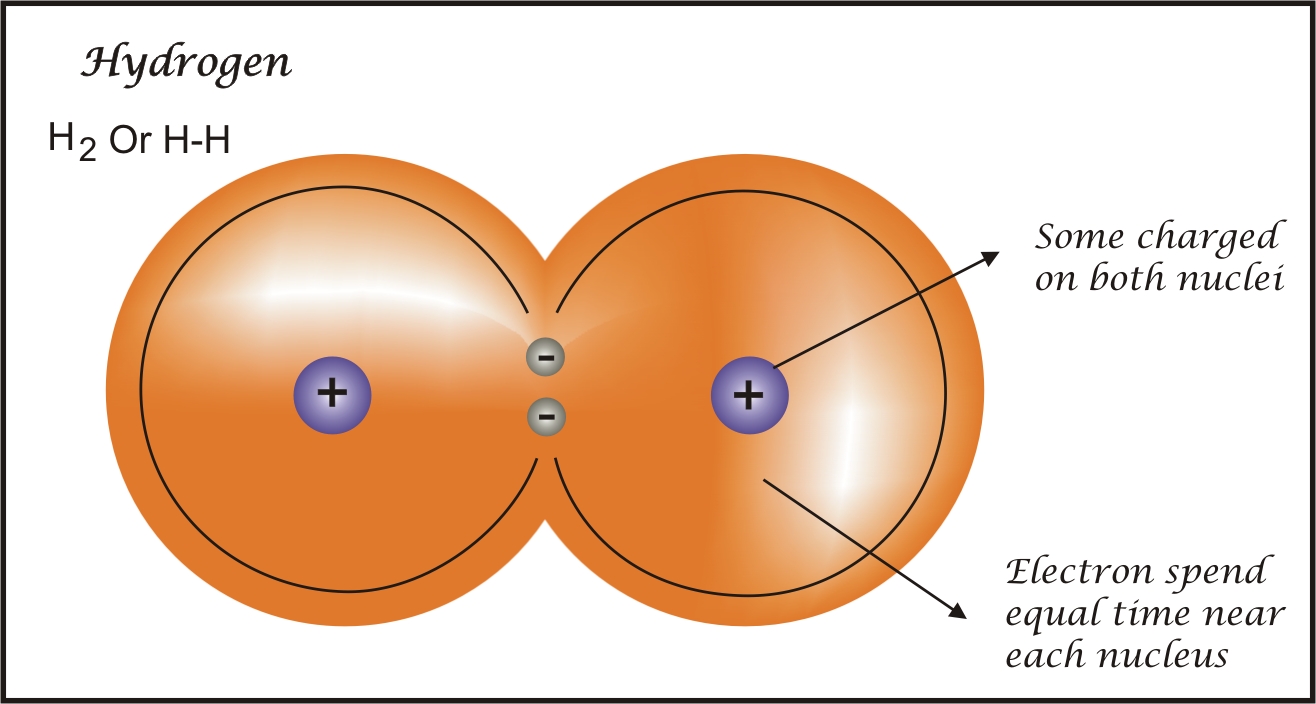
1.2. Polar Covalent Bonds:
-
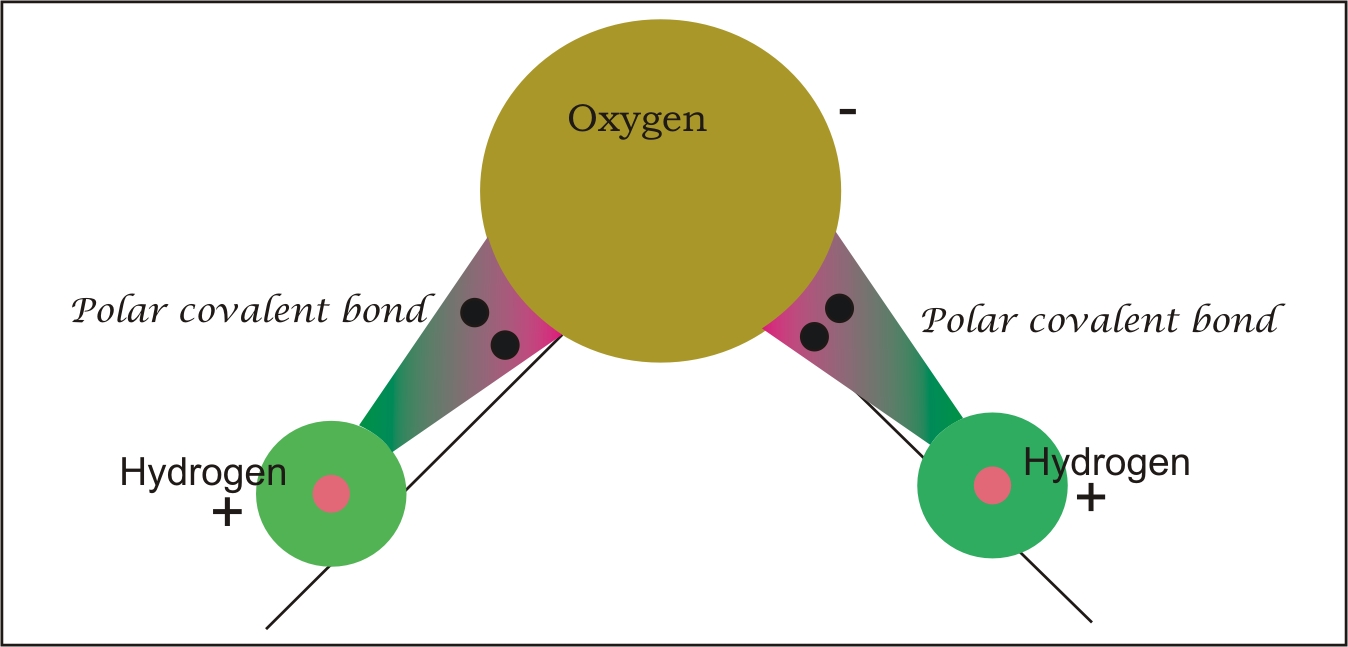
Due to the difference in the electronegative values of the atoms, the sharing of electrons become unequal.
-
The bond formed due to an unequal sharing is called a polar covalent bond.
-
Herein, one atom is partially electronegative and the other partially electropositive.
-
For example, H2O
2. Ionic Bonds:
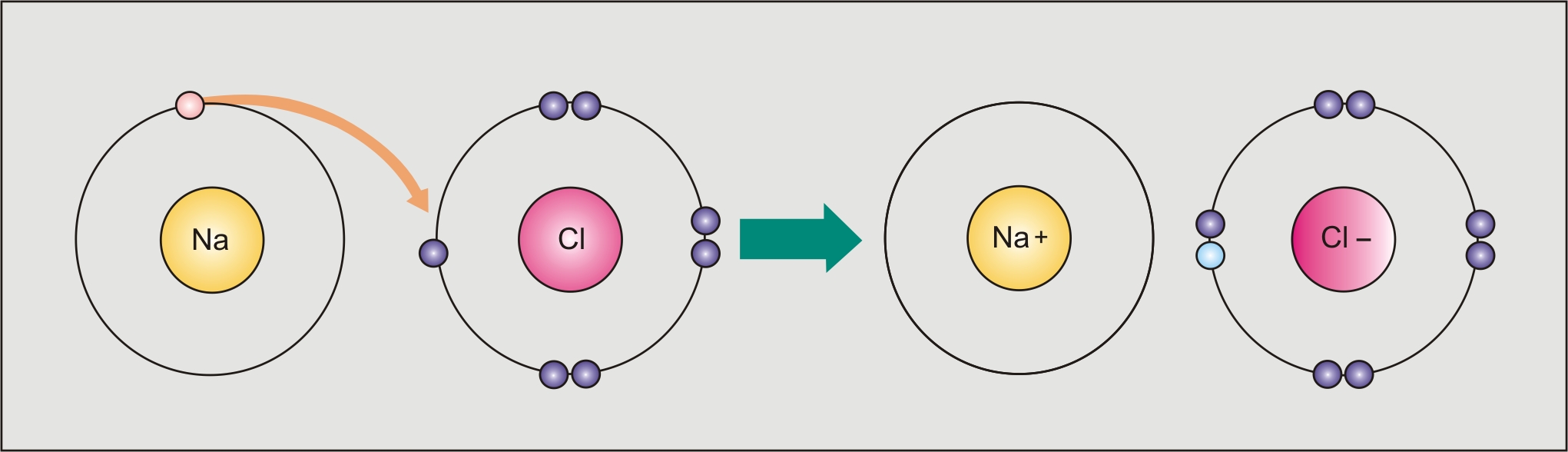
- These bonds are formed between two oppositely charged ions.
- One is more electronegative such that the electrons are completely transferred from the electropositive atom.
Intermolecular Bonds:
- The bonds between the atoms of different molecules are called the intermolecular bonds. These are of following types:
1. Hydrogen Bonds:
- When hydrogen is shared between two electronegative atoms, the hydrogen bond is formed. These are the strongest types of intermolecular bonds.
- The molecule which has the H-atom is called the H-bond donor while the molecule which accepts the H-bonds is called the H-bond acceptor.

2. London-Dispersion Forces:
- The bonds that are formed due to fluctuating electron densities around the molecules are called London-Dispersion forces.
Study other Related Concepts
Intramolecular and Intermolecular Bonds Current Topic
"Stay in the loop. Receive exam news, study resources, and expert advice!"
Get Answer to all your questions
Explore on Careers360
JEE Main
RPVT
Colleges By Branches
Colleges By Exam
Colleges By Branch
Colleges By Exams
Colleges By Ownership
Colleges By State
Colleges By Exams
Colleges By Degree
Colleges by State
Colleges by City
Colleges by State
Universities by Branches
By State
Colleges by City
Colleges by State
By State
BE/B.Tech
Diploma
MBA Specialization Colleges
Student Community: Where Questions Find Answers
