JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Preparation of Aldehydes is considered one the most difficult concept.
34 Questions around this concept.
Ethanol upon treatment with which one of the following will give ethanal?
Consider the following reaction:

Compound $X$ on reaction with $\mathrm{O}_{3 \text { followed by }} \mathrm{Zn} / \mathrm{H}_2 \mathrm{O}_{\text {gives formaldehyde }}$ and 2-methyl propanal as products. The compound $X$ is :
Re-NEET 2026: 30-Day Study Plan PDF | Last 5 Year's PYQ's with Solutions
Re-NEET 2026: NEET Previous 17 Years QP (2009–2025) | Most Scoring Concepts
Re-NEET 2026: Crash Course for Re-NEET 2026 | 100% FREE | Mock Test
Ozonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of
One mole of a symmetrical alkene on ozonolysis gives two moles of an aldehyde having a molecular mass of 44 u . The alkene is
Cemmensen reduction of a ketone is carried out in the presence of which of the following?
The best reagent to convert pent-3-en-2-ol into pent-3-en-2-one is
Oxidation
Oxidation of ROH involves the cleavage of (O-H) and (C-H) bonds to form (C=O) bond. Such reactions are also called dehydrogenation reactions because H2 is lost from an alcohol molecule. Strong oxidising agents such as acidic KMnO4 or acidic K2Cr2O7 oxidise 1o ROH first to an aldehyde and then to carboxylic acids. Further, 2o alcohols are oxidised to ketones and 3o alcohols do not undergo oxidation, but under drastic conditions such as strong oxidising agents(KMnO4) and at high temperatures cleavage of (C-C) bonds takes place and a mixture of RCOOH containing lesser number of C atoms is formed. 1o ROH can be converted upto aldehyde stage either by the use of CrO3 in anhydrous medium or with PCC or with Jones reagent.
Some examples include:

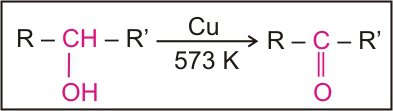
Dehydrogenation
1o alcohols undergo dehydrogenation to give aldehydes only. 2o alcohols give ketones and 3o alcohols give alkenes. Some examples include.

Rosenmund Reduction
Partial hydrogenation of benzoylchloride with finely divided Pd as catalyst in the prsence of BaSO4 and S or quinoline in boiling xylene as solvent gives benzaldehyde. This reaction is called as Rosenmund reduction. The catalyst under the above condition is called Lindlar's catalyst or poisoned Pd. The Lindlar's catalyst also reduces (C C) to (C=C) in syn-addition.
It is BaSO4 that prevents the aldehyde from being further reduced to alcohols and acts as a poison to the Pd catalyst. Small amount of sulphur and quinoline is very effective in poisoning the catalyst in aldehyde reduction. Moreover, S and quinoline react with small amount of H2 to give H2S gas and hydroquinoline, thereby limiting H2 for further reduction of aldehyde to alcohol.
The reaction occurs as follows:

Stephen Reduction
Nitriles are partially reduced to corresponding imine with SnCl2 in the presence of HCl, which on hydrolysis gives corresponding aldehyde. It does not reduce (C=C) or (C C). This reaction is known as Stephen reduction. The reaction occurs as follows:

From Acyl chlorides
Formyl chloride gives an aldehyde and all other halides five ketone. The reaction occurs as follows:

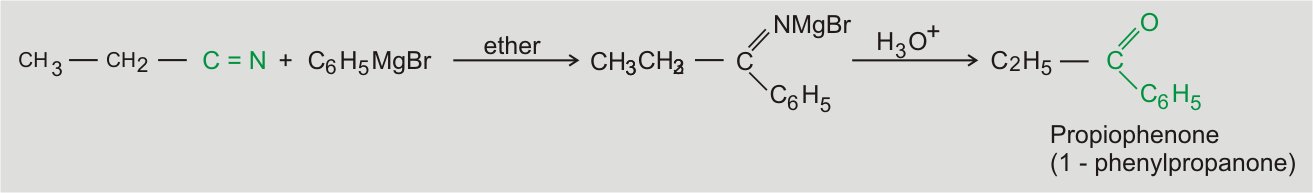
From Nitriles
Grignard reagents give aldehydes with hydrogen cyanide (HCN) and ketones with alkyl cyanides (RCN). The reaction occurs as follows:
"Stay in the loop. Receive exam news, study resources, and expert advice!"