Amrita Vishwa Vidyapeetham Allied & Life Science 2024
Admissions Open for multiple allied and health sciences programs across 5 campuses | Ranked #7 in India by NIRF, NAAC A++ Accredited
Instantaneous Rate of Reaction is considered one the most difficult concept.
Factors Affecting Rate of Reaction, Factors Affecting Rate of Reaction(2) is considered one of the most asked concept.
49 Questions around this concept.
Consider the reaction
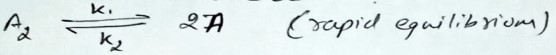
(slow)
If we increase the concentration of two times, then the rate of formation of AB will:-
Consider the reaction
the energy profile diagram for the reaction is given below:-

Question : what is the enthalpy of reaction .
Consider the reaction
the energy profile diagram for the reaction is given below:-

Question: Threshold energy of the reactant is
For the reaction, , the rate equation can be expressed in two ways
and
are related as :
For the chemical reaction,
the correct option is:
In the reaction
The rate of appearance of bromine (Br2) is related to rate of disappearance of bromide ions as following:
For the reaction, the value of the rate of disappearance of N2O5 is given as
. The rate of formation of
and
is given respectively as:
Consider the reaction, products. When concentration of
alone was doubled, the half-life did not change. When the concentration of
alone was doubled, the rate increased by two times. The unit of rate constant for this reaction is
The rate law for a reaction between the substances A and B is given by rate =
On doubling the concentration of A and halving the concentration of B, the ratio of the new rate to the earlier rate of the reaction will be as
It is defined as "The rate of change of concentration of a reactant or a product per unit time" that is it is nothing but it is the per unit mole of rate of decomposition of any reactant or formation of product.
As rate of reaction varies greatly with time, so generally, average reaction rate and instantaneous reaction rates are used. Average rate ways to is the rate at some particular instant of time and is equal to the time rate of change of the active mass of any of the reactants or any of the products.


Change in the concentration of reactants or products per unit time is called average reaction velocity. If Δc is the change in the concentration of reactants and product in Δt time, then:

As average reaction rate fails to predict rate at a particular moment of time so we use instantaneous rate which is equal to small change in concentration (dx) during a small interval of time (dt). It is given as dx/dt.

There are various factors on which the rate of reaction depends:

"Stay in the loop. Receive exam news, study resources, and expert advice!"
