JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
The National Testing Agency (NTA) conducts the NEET UG once a year. Due to paper leak allegations, the NEET 2026 exam has been cancelled by the NTA which was conducted on May 3, 2026. The single national-level undergraduate medical entrance exam, NEET UG 2026, will be conducted again in offline mode for admission to 645 medical, 318 dental, 914 AYUSH, and 47 BVSc and AH colleges in India.
The NTA has activated the NEET refund link 2026 at neet.nta.nic.in. Candidates can submit their bank account details and claim their exam fee refund till May 27, 2026. The revised NEET exam date 2026 is June 21, 2026. Candidates are not required to register again for the re NEET 2026 exam.
The NEET syllabus 2026 includes topics from classes 11 & 12 from Physics, Chemistry and Biology subjects. A total of 180 questions will be asked in NEET 2026 exam. Candidates need to answer these 180 questions within a time duration of 3 hours. These 180 questions are divided into 3 sections - Physics, Chemistry, and Biology. For every correct answer, 4 marks are given, whereas one mark is deducted for every wrong answer, and no marks are given for any unanswered questions.

| Full Exam Name | National Eligibility cum Entrance Test |
| Short Exam Name | NEET |
| Conducting Body | National Testing Agency |
| Frequency Of Conduct | Once a year |
| Exam Level | National Level Exam |
| Languages | Assamese +12 more |
| Mode Of Application | online |
| Application Fee | Online : 1700 |
| Mode Of Exam | offline |
| Mode Of Counselling | online |
| Participating Colleges | 2364 |
| Exam Duration | 3 Hours |
| No Of Seats | 178484 |
NEET National Eligibility cum Entrance Test (session 2026)
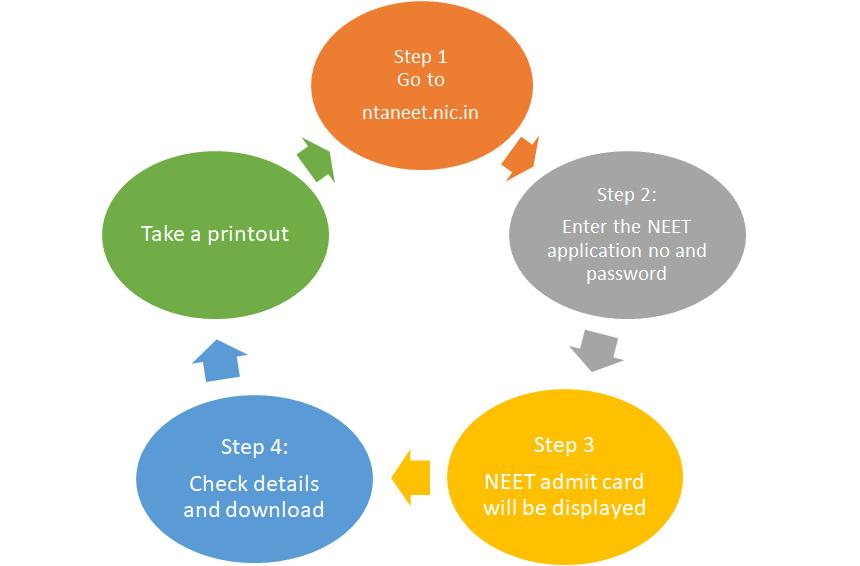
Registered candidates can download the admit card for NEET at neet.nta.nic.in. Carrying NEET 2026 admit card is mandatory on the exam day to verify the candidate's identity. Steps to download the admit card of NEET 2026 are as follows:
How to download the NEET admit card 2026?
Step 1: Click on the NEET 2026 admit card download link provided on the website
Step 2: Enter the NEET application number, date of birth and security pin
Step 3: NEET admit card will be displayed
Step 4: Download the admit card and check the details
Step 5: Print the same if the details are correct.
NEET 2026 admit card details
Important Tip: Candidates must paste a photograph in the space given on the admit card for the NEET exam. They must also put their left thumb impression and get their parent's signature in the space provided. Candidates must put their signatures in front of the invigilator at the NEET exam hall.
Image of NEET admit card

What to carry along with the NEET admit card 2026?
NEET 2026- how to correct the discrepancy in admit card?
Candidates must approach the NTA NEET helpline to rectify any discrepancy. They must email NTA with proof and the details of corrections required. Candidates will be allowed to appear for the exam with the old admit card. NTA will rectify and issue the correction at a later stage. NTA NEET helpline details are
What is the dress code for NEET 2026?
NEET exam 2026: Dress code for male candidates
NEET 2026: Dress code for female candidates
Customary dress code for NEET - Certain religions prescribe a customary dress code. Students who belong to such religions must have indicated in their application for NEET. For example, Sikh candidates can carry traditional Kangha, Kara, and Kirpan with them to the NEET 2026 exam hall.
NEET exam day instructions
Candidates must follow the given guidelines during NEET 2026 exam:
Candidates must sign the attendance sheet and admit card of NEET 2026 in front of the invigilator.
They must submit the photograph to be pasted on the NEET exam attendance sheet.
Candidates must read and understand all the instructions mentioned on the NEET question paper
All details in the particulars section of the NEET OMR sheet must be filled in legible handwriting. Care must be taken to ensure details are correct.
Candidates must not talk to other candidates and should maintain silence during the NEET exam.
They must not indulge in malpractices and avoid carrying objects, gadgets and other things that are barred from the NEET 2026 exam.
With the COVID pandemic threat still in place, candidates must also follow social distancing, wear masks and carry sanitiser to the exam hall for NEET 2026.
The exam will be video graphed so candidates must ensure they sit upright and avoid any suspicious activity or behaviour.
Candidates appearing for the NEET 2026 exam must meet the NEET 2026 eligibility criteria set by the NTA.
| Nationality | Indian Nationals, NRIs, PIOs, OCIs and Foreign Nationals |
| NEET 2026 Age Limit | Lower limit:17 years plus as of December 31, 2026 Upper age Limit: None |
| Qualifying exam details required for NEET | Class 12 or equivalent with PCB. |
| Class 12 percentage required for NEET 2026 | Gen/ OBC: Minimum 50% in PCB and aggregate SC/ST: Minimum 40% PwD: 40% |
NEET attempt limit | No limit |
| Class 12 Board | Students from recognised boards including NIOS and open schools |
| Biology as an extra subject | Allowed |
| Course | Class 12 percentage required for NEET |
| MBBS/ BDS | Minimum 50% in class 12 (40% for SC/ST and 40% for PWD). |
| BUMS | In addition to the above, pass in Urdu or Arabic or Persian language in class 10. |
| BSMS | Pass in Tamil subject in class 10 or equivalent in addition to class 12 criteria given above. |
The steps to fill out the NEET 2026 application form are as follows:
NEET 2026 application form fee
| Category | Fee + GST |
| General | Rs 1700 |
| OBC NCL/ Gen -EWS | Rs 1600 |
| SC/ST/Pwd/Transgender | Rs 1000 |
Important Tip: Keep 6 to 8 photographs for NEET 2026 exam day attendance sheet, admit card and medical admissions.
To correct any mistakes made in the application form, candidates can use the NEET correction window. Except the mobile number and email address, changes and corrections are allowed through NEET candidate login. The correction window of the NEET 2026 exam is a one-time facility and no chance is provided in future.
Corrections allowed in NEET application form 2026:
Practice with the NEET 2026 Free Mock Test PDF featuring full-length ReNEET exam simulation, detailed solutions, and real exam pattern.
Try Now| State | City |
| Andaman and Nicobar Islands | Port Blair |
| Andhra Pradesh | Guntur Kakinada Kurnool Nellore Rajahmundry Tirupati Vijayawada Visakhapatnam Vizianagaram |
| Assam | Dibrugarh Guwahati Silchar Tezpur |
| Bihar | Gaya Patna |
| Chandigarh | Chandigarh |
| Chhattisgarh | Bilaspur Raipur |
| Dadra and Nagar Haveli | Dadra and Nagar Haveli |
| Daman and Diu | Daman |
| Delhi | Central Delhi East Delhi North Delhi South Delhi West Delhi |
| Goa | Panaji |
| Gujarat | Ahmedabad Anand Bhavnagar Gandhinagar Godhra Patan Rajkot Surat Vadodara Valsad |
| Haryana | Faridabad Gurgaon |
| Himachal Pradesh | Dharamshala Hamirpur Shimla |
| Jammu and Kashmir | Jammu Srinagar Udhampur Leh Ladakh |
| Jharkhand | Bokaro Jamshedpur Ranchi |
| Karnataka | Bangalore Belgaum Davanagere Dharwad Gulbarga Hubli Mangaluru Mysore Udupi |
| Kerala | Alappuzha Ernakulam Kannur Kollam Kottayam Kozhikode Malappuram Palakkad Thrissur Thiruvananthapuram |
| Madhya Pradesh | Bhopal Gwalior Indore Jabalpur Ujjain |
| Maharashtra | Ahmednagar Amravati Aurangabad Beed Buldana Jalgaon Kolhapur Latur Mumbai Mumbai Suburban Nagpur Nanded Nashik Pune Satara Solapur Thane |
| Manipur | Imphal |
| Meghalaya | Shillong |
| Mizoram | Aizawl |
| Nagaland | Dimapur Kohima |
| Odisha | Angul Balasore Cuttack Rourkela Sambalpur Bhubaneswar Berhampur |
| Puducherry | Puducherry |
| Punjab | Amritsar Bathinda Jalandhar |
| Rajasthan | Ajmer Bikaner Jaipur Jodhpur Kota Udaipur |
| Sikkim | Gangtok |
| Tamil Nadu | Chennai Coimbatore Kancheepuram Madurai Namakkal Salem Thiruvallur Tiruchirappalli Tirunelveli Vellore |
| Telangana | Khammam Hyderabad Warangal K.V.Rangareddy |
| Tripura | Agartala |
| Uttarakhand | Dehradun Haldwani Roorkee |
| Uttar Pradesh | Allahabad Bareilly Ghaziabad Gorakhpur Jhansi Kanpur Lucknow Meerut Noida Varanasi |
| West Bengal | Burdwan Durgapur Hooghly Howrah Kharagpur Kolkata Siliguri North 24 Parganas |
| Arunachal Pradesh | Itanagar Naharlagun |
| Lakshadweep | Kavaratti |
Before looking at the NEET exam pattern, students should check the syllabus. NEET syllabus is the 10+2 level for Physics, Chemistry and Biology. Students must not only complete the NEET syllabus on time but also know what study material to choose. In addition, the best books for NEET ought to be close by such that students can refer and prepare for NEET 2026 in a better manner.
| Salient Factors | Details |
| Mode of Exam | Pen and Paper-based (Offline) |
| Duration | 3 hours |
| Language | English, Hindi, Assamese, Bengali, Gujarati, Kannada, Marathi, Odia, Tamil, Telugu, Urdu, Punjabi and Malayalam. |
| Subjects | Biology (Botany+Zoology), Physics and Chemistry |
| Level of NEET questions | 10+2 |
| Total Questions asked | 180 |
| Total Marks | 720 |
| Type of Questions | MCQs |
| Marks for a correct answer | 4 marks |
| Negative marking | -1 mark |
NEET schedule | Date and Time |
Duration of NEET Exam | 2:00 PM to 5: PM (3 hours) |
Last entry in the examination hall for NEET 2026 | 1:30 PM |
Sitting in the NEET exam room | 1:15 PM |
Announcement instructions and verifying NEET admit cards | 1:30 PM to 1:45 PM |
Distribution of the NEET 2026 test booklet | 1:45 PM |
Writing of particulars on the Test Booklet by the candidates | 1:50 PM |
Commencement of NEET examination | 2:00 PM |
NEET exam 2026 concludes | 5:00 PM |
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Physics: Unit 01
Physics: Unit 02
Physics: Unit 03
Physics: Unit 04
Physics: Unit 05
Physics: Unit 06
Physics: Unit 07
Physics: Unit 08
Physics: Unit 09
Physics: Unit 10
Physics: Unit 11
Physics: Unit 12
Physics: Unit 13
Physics: Unit 14
Physics: Unit 15
Physics: Unit 16
Physics: Unit 17
Physics: Unit 18
Physics: Unit 19
Physics: Unit 20
Chemistry: Unit 01
Chemistry: Unit 02
Chemistry: Unit 03
Chemistry: Unit 04
Chemistry: Unit 05
Chemistry: Unit 06
Chemistry: Unit 07
Chemistry: Unit 08
Chemistry: Unit 09
Chemistry: Unit 10
Chemistry: Unit 11
Chemistry: Unit 12
Chemistry: Unit 13
Chemistry: Unit 14
Chemistry: Unit 15
Chemistry: Unit 16
Chemistry: Unit 17
Chemistry: Unit 18
Chemistry: Unit 19
Chemistry: Unit 20
Zoology: Unit 01
Zoology: Unit 02
Zoology: Unit 03
Zoology: Unit 04
Zoology: Unit 05
Zoology: Unit 06
Zoology: Unit 07
Zoology: Unit 08
Botany: Unit 01
Botany: Unit 02
Botany: Unit 03
Botany: Unit 04
Botany: Unit 05
Botany: Unit 06
Botany: Unit 07
One important exercise in NEET preparation is the use of mock tests. One can only perfect what one has learned by practicing with past year question papers, mock tests, and such. Candidates can practice with these mock tests of NEET either on the NTA website or they can even download the tests to practise at home. NTA has also facilitated practising on NEET sample papers at TPCs which are the 2900 plus test practice centres set up at the nearest colleges in most towns and cities.
Candidates wishing to practice at the NEET TPCs must register and book the slot and go to the college to practise. The questions asked in these mock tests for NEET will be based on the past question papers of the exam.
The mock tests are only for practice hence scores are not given out. However, the feel and look of the actual exam can be gauged with the help of these tests.
Preparing for the NEET is the first step and this starts in class 11 for most students aspiring to appear for the exam. With the fierce competition and fewer seats, qualifying with a very good NEET score requires the best efforts. Effective preparation tips to crack the NEET exam include some basic guidelines and a good strategy. Since the aim is to crack NEET in the first attempt, some golden rules to follow are:
NEET exam 2026 strategy will also depend upon the following factors:
Tip 1: Understanding of NEET syllabus and topics- The question paper will have questions from Physics, Chemistry, and Biology at classes 11 and 12 levels. To crack NEET 2026, it is vital to know the topics to cover.
Tip 2: Strategise with Do and Die Chapters for NEET: The past year's question papers show questions asked from some topics while some topics are barely touched. These are mostly available online and also through a close look at the NEET question papers from past years.
Tip 3: Check NEET Exam Pattern - This will help candidates to know what the exam will be like and help in planning an exact NEET exam strategy.
Tip 4: Figure out Best Books for NEET 2026: Students ask whether NCERT books are enough to crack NEET exam? Since they help in cementing the foundation, NCERT books are important to prepare for NEET. However other books are also needed for reference.
In addition, some online resources for NEET Preparation are:
Tip 5: Schedule NEET Exam Timetable- the number of hours to study, what to study, and how to strategize is pretty important if one's goal is score 600 plus in NEET.
Tip 6: Tune in a Good NEET Revision Strategy: Just studying won't help. Revision of every chapter till one is thorough is a standard mantra for cracking the NEET exam.
Tip 7: Timely Testing and Evaluation: While intense preparation is necessary, it will be fruitful only if it is backed up by proper evaluation. This requires timely testing and proper revision to plug the gaps and holes in the preparation for the NEET exam 2026. One way of evaluation is the use of NEET Mock Tests and the NEET Previous Year Question Papers
Tip 8. Know from NEET Toppers: Well, NEET toppers from the previous year are good sources of knowing the pressures of exam preparation and how to succeed. Read their success mantras to know and tweak the NEET exam strategy.
Tip 9: Motivation and Determination: The path to success in NEET 2026 won't be easy but perseverance and determination are needed along with strong motivation to keep going and taste the sweetness of success.
Tip 10: Common Mistakes to Avoid in NEET exam: Well, these are common mistakes made by students and will have to be avoided. Check these out and know how to avoid them.
NEET Study Material 2026
To crack NEET UG 2026, students need to choose the best study material to plan their NEET preparation wisely. The NEET study material covers accurate, updated, necessary chapters and essential topics which helps students to cover the NEET 2026 syllabus and clear all major concepts from Physics, Chemistry and Biology. It includes all necessary, comprehensive resources which aim at a thorough understanding of facts and extensive preparation of NEET UG.
Key advantages of NEET study material 2026:
Enhance time management skills: With high quality NEET study material, it enables students to grasp complex concepts and enhance their speed and accuracy during the NEET exam.
Availability of study resources: The availability of question banks, modules and important books helps students to cover NEET syllabus by referring to well-structured study material.
Boost confidence: The comprehensive best study material for NEET enables students to prepare all sections of the exam with clarity and thus plays a crucial role in boosting confidence among students.
Keep updated with the latest trends: Aspirants can use the latest NEET material for NEET preparations. It keeps them updated with the latest NEET exam trends which helps them to figure out the necessary topics asked in the exam.
Quality content: The NEET study material is prepared by highly qualified professionals. The study material includes complete study material, detailed explanation of NEET topics, chapter-wise solved questions with high-quality content.
NTA will release the NEET 2026 provisional answer key on the official website. Candidates can use the NEET 2026 answer key by NTA to calculate their estimated scores and raise challenges by paying Rs. 200 per question. The final NEET UG 2026 answer key by NTA will be released along with the result and cannot be challenged further.
How to challenge NEET 2026 answer key by NTA?
Log in to the candidate portal
Check the provisional NEET answer key and OMR Sheet
Identify errors and discrepancies
Submit NTA NEET answer key challenge
Pay the NEET question challenge fee
Await response from the authorities
The previous year's NEET exam analysis helps candidates understand what the trends may be like. As per experts, the difficulty level of the previous year's NEET paper was moderate to difficult. While NEET physics paper was easy to moderate level, NEET biology and chemistry was of moderate to difficult level.
Numerical questions dominated the NEET physics paper with 78% which is more in comparison to the previous year. Questions in chemistry were based on the NCERT statements, facts and graphs. In Botany, the majority of questions are factual with a few concept-based questions. The zoology section is based on the NCERT syllabus and requires an understanding of the meaning between the lines
How to download NEET 2026 result?
NEET Scorecard 2026-Details mentioned
NEET Merit List 2026
For counselling and admissions, the NEET score and ranks are used to draw up the merit list. It is to be noted that the merit list of NEET 2026 will be used for seat allotment during counselling. Candidates must note that their result is the basis for the NEET merit list and this will be prepared after they apply for counselling and admissions for the All India and state admissions. It also must be noted that merit lists of the NEET exam will be separate for All India and state counselling.
To qualify for the test, candidates must score equal to or above the specified NEET cutoff 2026. This is the minimum percentile that is pre-decided by MCI and stipulated in the Graduate Medical Education Regulations (1997) as the score which a candidate must obtain to be eligible for admissions.
The NEET qualifying percentile has been specified category-wise and can be summarized as given in the table below. To aid candidates, the actual marks secured in the past years have also been mentioned such that candidates get an idea of how much to score to qualify for the exam.
The cutoff score is determined by the number of applicants, difficulty levels of the question paper, seats available, and past cutoff trends.
Category | Qualifying percentile | Cutoff score |
UR/EWS | 50th percentile | 686 - 144 |
OBC | 40th percentile | 143 - 113 |
SC | 40th percentile | 143-113 |
ST | 40th percentile | 143-113 |
UR/ EWS - PwBD | 45th percentile | 143 - 127 |
OBC PwD | 40th percentile | 126 - 113 |
SC PwD | 40th percentile | 126 - 113 |
ST PwD | 40th percentile | 126 - 113 |
The Medical Counselling Committee (MCC) conducts the counselling for NEET in the online mode including the choice filling and locking process. The registrations for the NEET counselling 2026 can be done before the last date. The result of the round-wise seat allotment process of the NEET counselling is declared online.
Candidates who are allotted seats in NEET UG 2026 counselling must report to the allotted college for document verification within a certain period with the NEET counselling documents. NEET seat allotment will be based on the All India Rank of the applicants, choice of colleges entered, seat availability, reservation criteria etc.
MCC NEET 2026 counselling is conducted in three rounds: Round one, Round two, Round 3, followed by a stray vacancy round. Candidates who qualify for NEET 2026 will be eligible to apply for the All India counselling. However, for the respective state counselling processes, admissions will be based on factors like domicile, residency in state etc. Candidates must check whether they meet the state admission criteria before applying for state counselling.
NEET counselling is of two types. One is the All India counselling conducted by DGHS for 15% All India Quota seats in government medical colleges and all seats in Central Institutions including AIIMS, JIPMER, AFMC, ESI, Delhi University (DU), BHU, AMU and Deemed Universities. The second is the state counselling held by each state medical council for the rest of the 85% of the seats in the government colleges, state-run universities and the private medical colleges of the state. Only students who qualify for NEET will be eligible for counselling subject to certain regulations.
MCC NEET Counselling guidelines
| Type of Counselling | Counselling Body |
Counselling for 15% AIQ Seats (except in Jammu & Kashmir) in Government medical and dental colleges | DGHS, on behalf of MCC |
| Counselling for all MBBS seats in AIIMs and JIPMER | |
Counselling for all the seats in Central and Deemed Universities including BDS at Jamia Millia Islamia, New Delhi | |
| Counselling for 85% State Quota seats in DU colleges (MAMC, LHMC, UCMS) | |
Counselling for IP Quota seats in ESIC Colleges | |
Counselling for Armed Forces Medical College (AFMC) | DGHS, on behalf of MCC and AFMC Pune |
Counselling for 85% State Quota seats and 100% state private colleges. | Respective State Counselling Authorities |
NEET State-wise Counselling
The question paper for NEET is published by the National Testing Agency (NTA) along with the OMR sheet and answer keys. Candidates can download the NEET question paper PDF by logging in with their credentials from the official website neet.nta.nic.in. To prepare better for the undergraduate medical exam, it is important for candidates to solve the NEET previous year's papers. Solving the previous year's questions will help aspirants have an idea of the NEET UG difficulty level.
| NEET question combined sets | English | Hindi |
| E1+F1+G1+H1 | Click here | Click here |
| E2+F2+G2+H2 | Click here | Click here |
| E3+F3+G3+H3 | Click here | Click here |
| E4+F4+G4+H4 | Click here | Click here |
| E5+F5+G5+H5 | Click here | -- |
| E6+F6+G6+H6 | Click here | -- |
| NEET question paper language | Link |
| Telugu | Click here |
| Gujarati | Click here |
| Marathi | Click here |
| Tamil | Click here |
| Bengali | Click here |
| Kannada | Click here |
| Odia | Click here |
| Urdu | Click here |
| Assamese | Click here |
Contact Number:
918076535482 , 917703859909
Frequently Asked Questions (FAQs)
The exam date of NEET UG 2026 was May 3, 2026. The re NEET 2026 date is June 21, 2026.
NEET 2026 application form was released on February 8, 2026. The NEET UG registration last date was extended till March 11, 2026.
Yes, the scores obtained by the candidates in Class 12 will be taken into consideration for the eligibility criteria for NEET UG 2026 exam.
Yes, compartment students can apply for the NEET exam 2026. But, they have to produce the 12th class pass certificate and need to obtain the required percentage of marks.
Aspirants fulfilling the eligibility criteria set by the NTA for NEET 2026 can fill the application form.
The Medical Counselling Committee (MCC) will conduct the NEET counselling 2026.
The total duration of NEET 2026 will be of 3 hours.
Yes, the NEET dress code 2026 will be decided by the NTA. The NEET UG 2026 dress code must be followed by every candidate to avoid any hassle on exam day.
Once allotted, the NEET UG exam centre 2026 cannot be changed.
There is no restriction on the number of attempts in NEET UG.
On Question asked by student community
Hello,
Yes, with 74.6% in CBSE Class 12 , you are eligible to apply for B.Sc Nursing at Rachna Institute of Nursing.
As for NEET , admission without NEET may be possible, but it depends on the college's admission rules for 2026. It's best to confirm this directly with the
Hello Dear Student,
Yes, you can become a physiotherapist in India without appearing for NEET. Admission to the Bachelor of Physiotherapy (BPT) programme in many colleges is based on Class 12 marks in Physics, Chemistry, and Biology (PCB) or through university-specific entrance examinations. Candidates should check the eligibility criteria and
Admissions for the BPT (Bachelor of Physiotherapy) course at Sree Anjaneya College of Paramedical Sciences usually begin around the same time as the Kerala paramedical admissions process. If you've completed Class 12 with Physics, Chemistry, and Biology and meet the minimum marks requirement, you're generally eligible to apply.
The annual
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
College Available: 794
College Available: 386
College Available: 268