Liquid State MCQ - Practice Questions with Answers
Quick Facts
-
26 Questions around this concept.
Solve by difficulty
Two droplets merge with each other and form a large droplet. In this process:
In a liquid, a force of 5N, a velocity gradient of du/dx, and a contact area of 20m2 are needed to sustain the flow of layers. What then is the significance of viscosity?
Concepts Covered - 0
Liquid State
Intermolecular forces are stronger in liquid state than in gaseous state. Molecules in liquids are so close that there is very little empty space between them and under normal conditions liquids are denser than gases. Molecules of liquids are held together by attractive intermolecular forces. Liquids have definite volume because molecules do not separate from each other. However, molecules of liquids can move past one another freely, therefore, liquids can flow, can be poured and can assume the shape of the container in which these are stored. In the following sections we will look into some of the physical properties of liquids such as vapour pressure, surface tension and viscosity.
Vapour Pressure
At a particular temperature, it is the pressure exerted by vapours over liquid surface when vapours are in equilibrium with liquid.
- Vapour pressure increases with increase of temperature.
- The variation of vapour pressure of liquid with temperature is given as
Here, A = constant, P = Vapour pressure of liquid, T = Temperature - The plot of log P vs 1/T will be in a straight line.
- The vapour pressure of H2O at 373 K is 76 cm.
- At critical temperature the meniscus between liquid and vapour disappears.
- Vapour pressure Extent of H-bonding: HF > H2O > NH3
- Heat of vaporization H-bonding example, HF > H2O > NH3
- The amount of heat needed to convert one gram of a liquid into its vapour at its B.P is known as heat or enthalpy or latent heat of vaporization.
Surface Tension
It is the force at right angles to the surface of a liquid along one cm or one-metre length of the surface.
- Units: Newton metre-1 or Nm-1, dyne cm-1
- Due to surface tension the surface area of the liquid decreases upto minimum. example. , Falling drops are spherical that is, minimum surface area for a given volume.
- Due to surface tension a liquid rises in capillary tube, water move upward In soil and walking of insects over water surface.
- Surface tension ∝ 1/Temperature
- At critical temperature surface tension is zero.
- The effect of temperature is given by Eotvos equation.
Here, K = constant, d = density, M = Molar mass
Viscosity
- Viscosity
It is the internal resistance of a liquid to flow which exists due to the relative motion between two layers. It decreases with increase of temperature. It is calculated as the force per unit area needed to maintain a velocity difference of unity between two parallel layers of liquid unit distance apart.
-
Laminar Flow
The liquid is considered to be consisting of molecular layers arranged one over the other. When the liquid flows over a glass surface then the layer of molecules immediately in contact with the glass surface is stationary with zero velocity. But layer immediately above it is not stationary but flows with some velocity. Further, the next layer above it flows still faster and this continues and the topmost layer of molecules flow with maximum velocity. So, this type of flow in which there is a gradual gradation in the velocities on passing from one layer to another is called laminar flow.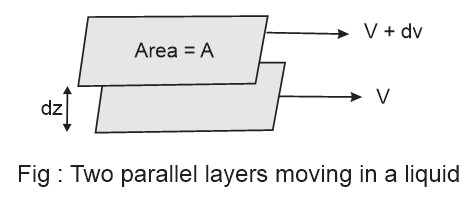
-
-
Viscosity Coefficient
It is the force of friction needed to maintain a velocity difference of 1 cm/sec between any two parallel layers of 1 cm2 area and which are 1 cm apart.
-
Effect of Temperature on Viscosity
On increasing temperature, viscosity decreases as average thermal energy of molecules increases hence the effect of intermolecular attraction forces decreases.
It can be shown by Arrhenius equation as follows:
Here T = Temperature, R = Universal gas constant, Ea = Activation energy -
Fluidity
It is the reciprocal of viscosity coefficient of a liquid denoted by 𝜙.
"Stay in the loop. Receive exam news, study resources, and expert advice!"
