Emversity Allied Health Programs
Get Job Ready in Healthcare | Employability-Focused Programs
8 Questions around this concept.
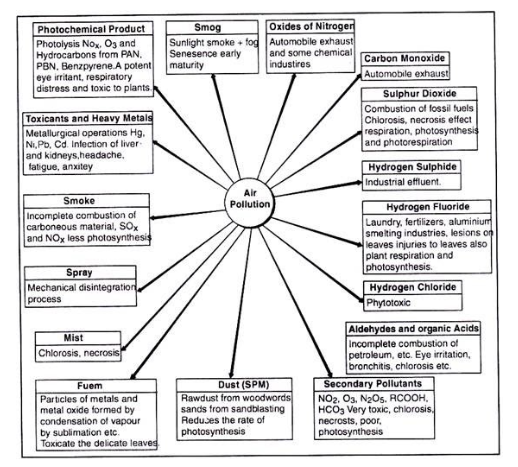
Which of the following is a secondary pollutant?
Which of the following statements is correct ?
1) Benzene hexachloride (BHC) is a non - biodegredable pollutant
2) Anthropogenic air pollutants are natural in origin
3) Carbon monooxide is a primary air pollutant
4) Sulphur dioxide causes brown air effect during traffic congestion in cities
Which one of the following statements is not valid for aerosols?
NEET 2026: Exam Centres List | Free NEET Coaching & Study Material
NEET Prep: Mock Test | 10 Years PYQ's | Syllabus
NEET 2026: Boards Cheat Sheet | Mind Maps & Diagrams Guide | Formula Sheet
Latest: Allied and Health Sciences | Paramedical Universities Accepting Applications
According to the Central Pollution Control Board, the diameter of particles that are responsible for causing great harm to human health are:
Among the following which one causes maximum indoor chemical pollution?
CNG is preferred as a fuel over diesel for public transport because
(i) It is cost-effective.
(ii) it burns almost completely.
(iii) it can be recycled.
(iv) it burns only partially.
Choose the correct combination.
Read the given statements and select the correct option.
Statement 1: MTP is considered relatively safe during the first trimester of pregnancy.
Statement 2: Foetus becomes intimately associated with the maternal tissues after the first trimester.
1. Gases:
2. Particulate Matters:
3. Secondary Pollutants:
"Stay in the loop. Receive exam news, study resources, and expert advice!"