JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Preparing for NEET 2026 is not just about covering the syllabus - it is about revising smartly and solving questions efficiently. One of the most important aspects of last-stage preparation is mastering key Physics and Chemistry formulas. The revision with the NEET formula sheet 2026 helps you solve numerical and concept-based questions quickly, which is important in a time-bound exam like NEET.
This Story also Contains

However, simply memorising formulas is not enough. You need to understand how they are applied in real exam questions. In Physics, formulas related to mechanics, electrostatics, thermodynamics, and Modern Physics are repeatedly asked. In Chemistry, formulas from physical chemistry, such as the mole concept, thermodynamics, equilibrium, and electrochemistry, are important. Learning these formulas in a structured way increases confidence and reduces mistakes. Thus, mastering NEET Physics and Chemistry formulas is an important step in NEET exam preparation.
| Important Physics Formula for NEET 2026 | Download Here |
The important physics formulas for NEET based on the NEET exam trends observed in the previous years are provided here. The table consists of the highest-scoring concepts in the last 5 years, the number of times they appeared in NEET, and the years in which questions were based on these concepts. The physics NEET 2026 formula sheet is based on these high-scoring concepts.
Also read:
NEET 2026 Exam Analysis PDF: Difficulty Level, Student Reactions, Subject-Wise Paper Review | |
NEET 2026 Physics Answer Key with Solutions PDF – Download | |
NEET 2026 Chemistry Answer Key with Solutions PDF – Download | |
NEET 2026 Biology Answer Key with Solutions PDF – Download | |
NEET 2026 Code 11 Question Paper with Answer Key & Solutions PDF – Download | |
NEET 2026 Code 12 Question Paper with Answer Key & Solutions PDF – Download | |
NEET 2026 Code 13 Question Paper with Answer Key & Solutions PDF – Download | |
NEET 2026 Code 14 Question Paper with Answer Key & Solutions PDF – Download |
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
S. No. | Concept title | Chapter | Number of times appeared in the Previous year's papers (last 6 years) |
1. | Series LCR circuit | Electromagnetic Induction and Alternating Currents | 6 |
2. | Equations of motion of SHM | Oscillations and Waves | 7 |
3. | Electric potential due to continuous charge distribution (II) | Electrostatics | 5 |
4. | Resistance and Resistivity | Current Electricity | 6 |
5. | Parallel Grouping of Resistance | Current Electricity | 4 |
6. | Kirchhoff's second law | Current Electricity | 4 |
7. | Nature of Electromagnetic Waves | Electromagnetic Waves | 4 |
8. | Total Internal Reflection | Optics | 4 |
9. | Young's double slit experiment -1 | Optics | 4 |
10. | De-Broglie wavelength of an electron | Dual Nature of Matter and Radiation | 4 |
11. | Logic Gates | Electronic devices | 7 |
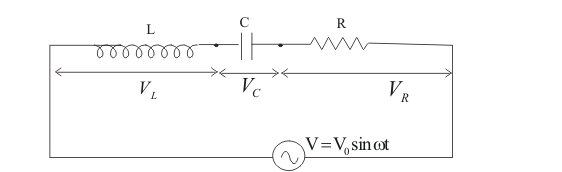
Let i be the amount of current in the circuit at any time, and VL, VC, and VR the potential drops across L, C, and R, respectively.
vR=iR⇒ Voltage is in phase with i,
vL=iωL⇒ Voltage leads i by 90∘,
vC=iωC⇒ Voltage lags i by 90∘.
By all these, we can draw a phasor diagram as shown below –
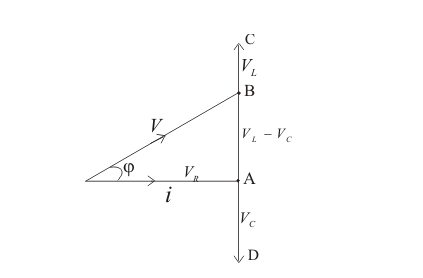
So, from the above phasor diagram, V will represent the resultant of vectors VR and (VL−VC).
So the equation becomes –
V=VR2+(VL−VC)2=(iR)2+(XL−XC)2=(iR)2+(ωL−1ωC)2=iZ,
where Z=R2+(ωL−1ωC)2 is called the impedance of the circuit.
Also, tanϕ=VL−VCVR=XL−XCR=ωL−1ωCR.
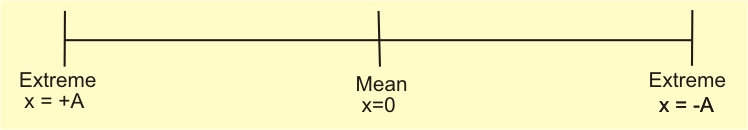
As we know, a=−ω2x
General equation of SHM
1. For Displacement:
x=Asin(ωt+ϕ) ; where ϕ is the initial phase and (ωt+ϕ) is called the phase.
Various displacement equations:
(1) x=Asinωt⇒ when the particle starts from mean position towards right.
(2) x=−Asinωt⇒ when the particle starts from mean position towards left.
(3) x=Acosωt⇒ when the particle starts from extreme position towards left.
(4) x=−Acosωt⇒ when the particle starts from left extreme position towards right.
2. For Velocity (v):
x=Asin(ωt+ϕ)⇒v=dxdt=Aωcos(ωt+ϕ)=Aωsin(ωt+ϕ+π2)
3. For Acceleration (a):
x=Asin(ωt+ϕ)⇒v=dxdt=Aωcos(ωt+ϕ)=Aωsin(ωt+ϕ+π2)
⇒a=dvdt=−Aω2sin(ωt+ϕ)=Aω2sin(ωt+ϕ+π)=−ω2x
So, the phase difference between x and v is π2.
Similarly, the phase difference between v and a is π2.
And, the phase difference between a and x is π.
Differential equation of SHM:
dvdt=−ω2x⇒ddt(dxdt)=−ω2x⇒d2xdt2+ω2x=0
If the motion of any particle satisfies this equation, then that particle performs Simple Harmonic Motion (SHM).
Based on the NEET 2026 exam pattern seen over the previous five years, we have assembled the important chemistry formulas for the NEET exam. The concepts that have scored the highest over the past five years, along with the number of times they have appeared and the years in which questions based on them have been asked, are listed in the table below. The NEET chemistry formula listed below is based on these high-scoring concepts:
S. No. | concept title | Chapter | Number of times appeared in the Previous year's paper (last 6 years) |
1. | Shapes of Molecules | Chemical Bonding and Molecular Structure | 3 |
2. | Solubility and Solubility Products | Equilibrium | 3 |
3. | The Gas Laws- Boyle’s Law (Pressure- Volume Relationship) | States of Matter: Gases and Liquids | 3 |
4. | Mathematical Analysis of Cubic System | Solid state | 4 |
5. | Charge on Colloids | Surface Chemistry | 3 |
6. | Oxidation State | d and f Block Elements | 3 |
7. | Preparation of Aldehydes | Aldehydes, Ketones and Carboxylic Acids | 4 |
8. | Nucleophilic Addition Reaction | Aldehydes, Ketones and Carboxylic Acids | 5 |
9. | Reduction and Oxidation reactions | Aldehydes, Ketones and Carboxylic Acids | 4 |
10. | Chemical Properties of Carboxylic Acids | Aldehydes, Ketones and Carboxylic Acids | 3 |
11. | Carbohydrates | Biomolecules | 3 |
Aspirants can download the important NEET chemistry formula from the table given below:
| NEET Chemistry Formula 2026 | Download Here |
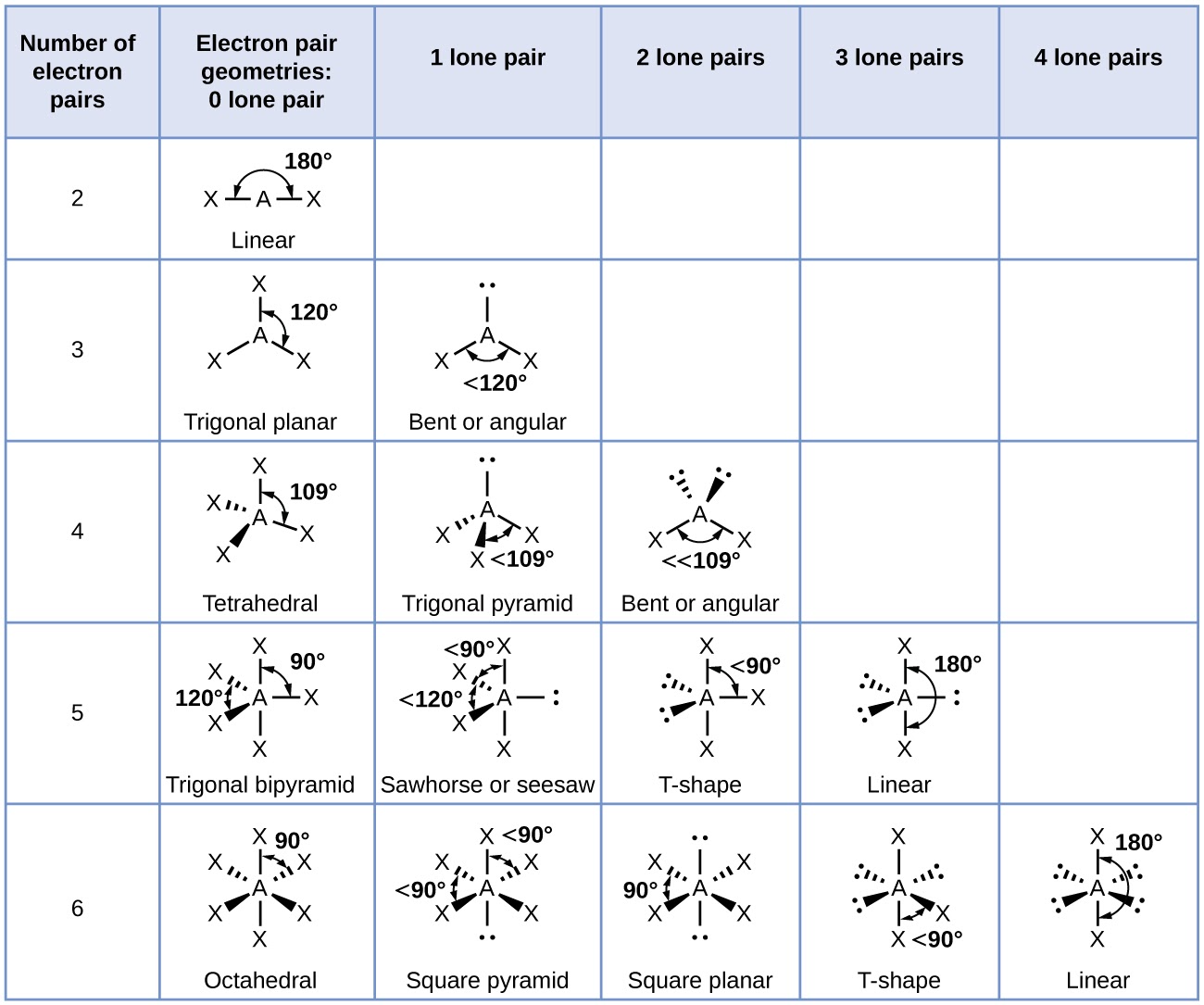
The ideal shapes of molecules, which are predicted based on electron pairs and lone pairs of electrons, are mentioned in the table below:
General Representation:
AxBy⇌xA++yB−
Ksp=[A+]x[B−]y
Relation between Solubility (s) and Solubility Product (Ksp):
AxBy⇌xA++yB−
If solubility is s, then [A+]=xs and [B−]=ys
Thus,
Ksp=(xs)x(ys)y=xxyysx+y
The Gas Laws — Boyle’s Law (Pressure–Volume Relationship):
P∝1V⇒PV=k(at constant T)
On Question asked by student community
Hello Dear Student,
Yes, qualifying for the NEET is mandatory if you are seeking admission to core medical courses like
MBBS
,
BDS
,
BAMS
,
BHMS
, and
BSc Nursing
across India.
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-eligibility-criteria
Hope it helps!
Hello Dear Student,
The NEET syllabus can seem overwhelming at first, but with a proper strategy and consistent effort, it becomes much more manageable. The key is to focus on the most important topics and practice regularly.
Effective NEET Preparation Tips
Prioritize NCERT Books: NCERT should be your primary study
Hello Dear Student,
No, NEET is not compulsory for Bachelor of Physiotherapy (BPT) admissions for the 2026–27 academic session.
You can check, find and access more information here:
https://news.careers360.com/physiotherapy-neet-admission-1yr-internship-bpt-dr-title-allied-health-sciences-paramedical-courses-revamp-syllabus-ncahp-mohfw
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-mds-2025-seat-allotment-result
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-2024-marks-vs-rank
https://medicine.careers360.com/articles/neet-2024-cut-off
Hope it helps!
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs