JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs

Chemical Kinetics is one of the important chapters of class 12 Chemistry. It generally deals with understanding the rate of chemical reactions. Rate of chemical reaction is the speed at which chemical reaction takes place. Reaction rate is influenced by many factors including temperature, pressure, catalyst etc.
Reactions are classified in various orders like zero order reaction, First order reaction, second order reaction among others on the basis of rate law followed by them.
A first-order reaction refers to the reaction in which the rate of reaction is proportional to the first power of concentration of the reactant at any point of time. Hence, higher the concentration of reactant, higher will be the rate of reaction. Rate of first order reaction decreases as it proceeds due to decrease in concentration of reactant.
Hence in first order reaction, rate of reaction is directly proportional to the concentration of reactant as described in the equation below.
Reaction-
Reactant →Product
Rate =k[Reactant]
Below are some of the examples of First Order reaction
2N2O5 → 4NO2+O2
Rate = k[N2O5]
2H2O2 → 2H2O + O2
Rate = k[H2O2]
After analysing the last five years of NEET papers, it has been observed that, total 10 questions were asked from the Chemical Kinetics Chapter and out of those, four questions belong to First order reaction. This shows that First order reaction is the most important concept of Chemical Kinetics and most of the questions can be solved if Integral form of first order reaction is understood well. Questions asked from first order reactions are discussed in the article below.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Differential Rate Law for a First-Order Reaction reflects the change in concentration with respect to time which is shown by equation below:
Let us assume a first order reaction: A(Reactant)→B(Product)
Rate = -d[A]/dt = k[A]1 = k[A]
In the above equation,
‘k’ represents the rate constant of the first-order reaction, the unit of the rate constant is s-1.
‘[A]’ represents the concentration of the reactant.
d[A]/dt represents the change in the concentration of reactant ‘A’ with respect to time.
Integrated Rate Law of first order reaction is used to calculate the value of rate of rate constant, concentration of reactant a given time, half life etc. Most of the numericals are solved by using Integrated rate law only. Differential rate law is used to obtain the Integral rate law of a First order reaction.
Differential rate law is: -d[A]/dt = k[A]
![]() d[A]/dt = -k[A]
d[A]/dt = -k[A]
Integrating both sides, we get
![]()
Where [A]o is concentration of reactant at time t0 s, and [A] is the concentration of reactant at time t. We get,
![]()
![]()
Considering t0 =0, and solving the equation, we get
![]() ln[A] – ln[A]0 = -kt
ln[A] – ln[A]0 = -kt
![]() ln[A] =ln[A]0 -kt
ln[A] =ln[A]0 -kt
Raising each side to component e, we get
![]()
![]()
![]()
![]()
Hence, ![]() is the final expression in the integrated form for first order reaction. The above derivation is useful in solving many numerical problems.
is the final expression in the integrated form for first order reaction. The above derivation is useful in solving many numerical problems.
Using the Integrated Rate law, Concentration v/s time Graph for First order reaction can be obtained which is shown below
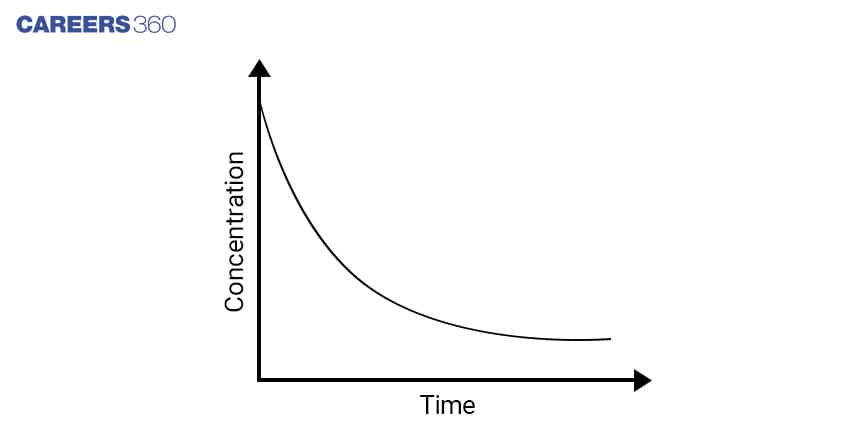
As the first-order reaction follows the equation ln[A] = ln[A]0 – kt which is similar to that of a straight line (y = mx + c) with slope -k. Hence, this equation can be plotted as-
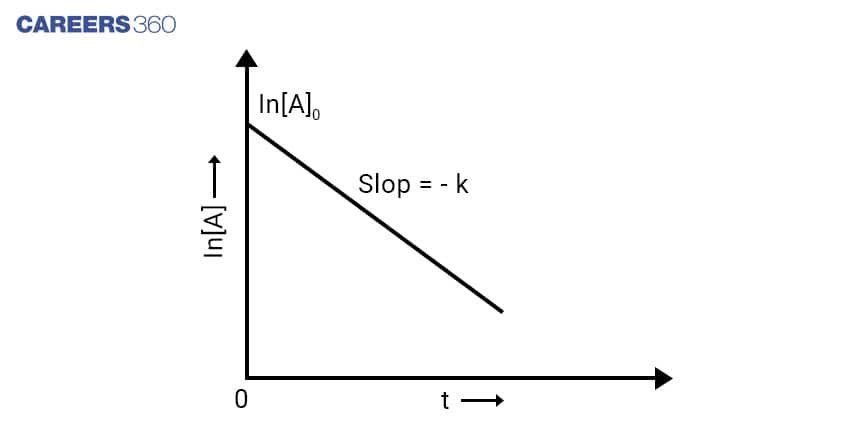
From the above graph, it can be said that the graph for ln[A] v/s t for a first-order reaction is a straight line with slope -k.
The half-life of a chemical reaction refers to the time required by the reactant concentration to become half of its initial value. It is represented by ![]() .
.
Hence, according to the first order reaction, ![]() . Where [A] refers to the concentration of the reactant at any time ‘‘t’’ and [A]o represents the initial concentration of the reactant.
. Where [A] refers to the concentration of the reactant at any time ‘‘t’’ and [A]o represents the initial concentration of the reactant.
At half life, t = t1/2 , [A] =A0/2
Putting the value of [A] =A0/2 and t = t1/2 in the equation ![]() , we get
, we get
![]()
![]()
Taking ln on both sides, we get,
![]()
![]()
Hence half life of the first order reaction is 0.693/k, where k is the rate constant.
Below discussed are some of the questions asked in the NEET exam from first order reaction.
Question 1. A first order reaction has a specific reaction rate of 10-2 sec-1. How much time will it take for 20 g of the reactant to reduce to 5 g? [NEET 2017]
238.6 sec
138.6 sec
346.5 sec
693.0 sec
Ans. The rate of first order reaction is proportional to the first power of the concentration of the reaction.
Let us consider a reaction, R → P , wherein
R → P
At t=0 a 0
At t=t a-x x
![]()
![]()
After solving this equation, we get
![]()
Now, putting the values
![]()
t= 138.6 sec
Alternatively, this can be solved by the half life method. According to which after the first half life concentration becomes half and after the second half life, concentration becomes ¼ th of the initial concentration. Hence after two half life, the concentration becomes from 20g to 5g.
![]()
![]()
![]()
Now, 2t1/2= 69.3×2= 138.6 sec.
Hence option ‘’C’’ is correct.
Question 2. The correct difference between first and second-order reactions is that [NEET 2018]
a first-order reaction can be catalysed; a second-order reaction cannot be catalysed
the half-life of a first-order reaction does not depend on [A]o; the half-life of a second-order reaction does depend on [A]o
the rate of a first-order reaction does not depend on reactant concentrations; the rate of a second-order reaction does depend on reactant concentrations
the rate of a first-order reaction does depend on reactant concentrations; the rate of a second-order reaction does not depend on reactant concentrations
Ans. For first Order reaction, rate is directly proportional to the first power of concentration.
For a reaction, A→B
Rate = -d[A]/dt = k[A] and half life is
![]()
Where unit of ![]() .
.
Hence ![]()
For second order reaction, For a reaction, A→B
Rate = ![]() and its half life is
and its half life is ![]()
![]()
Hence, The half-life of a first-order reaction does not depend on [A]o; the half-life of a second-order reaction does depend on [A]o
Option B is correct.
Question 3. If the rate constant for a first order reaction is k, the time (t) required for the completion of 99 per cent of the reaction is given by: [NEET 2019]
t = 0.693/k
t = 6.909/k
t = 4.606/k
t = 2.303/k
Ans. Let us consider a reaction, R → P , wherein
R → P
At t=0 a 0
At t=t a-x x
For the First Order reaction, Rate of reaction is proportional to the first power of the concentration of the reaction.
![]()
![]()
![]()
Unit of ![]()
![]() and
and
![]()
![]()
![]()
![]()
![]()
Hence option C is correct.
Question 4. The rate constant for a first order reaction is ![]() The time required to reduce 2 g of the reactant to 0.2g is: [NEET 2020]
The time required to reduce 2 g of the reactant to 0.2g is: [NEET 2020]
1000s
100s
200s
500s
Ans. Given,
![]()
Hence the given reaction is of first order.
Thus, we have:
![]()
Hence it is from the option of keeping th
![]()
Thus, ![]()
![]()
Therefore, Option(D) is correct.
On Question asked by student community
Hello Dear Student,
No, NEET is not compulsory for Bachelor of Physiotherapy (BPT) admissions for the 2026–27 academic session.
You can check, find and access more information here:
https://news.careers360.com/physiotherapy-neet-admission-1yr-internship-bpt-dr-title-allied-health-sciences-paramedical-courses-revamp-syllabus-ncahp-mohfw
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-mds-2025-seat-allotment-result
Hope it helps!
Hello Dear Student,
With a NEET MDS 2026 score of 503 and an AIR of around 4600, getting a government seat in highly competitive branches such as Orthodontics, Conservative Dentistry & Endodontics, or Prosthodontics may be difficult, as these specialties usually close at much better ranks.
However, you still have
Hello Dear Student,
Haryana NEET MDS 2026 counselling registrations are expected to begin in July 2026. The official schedule will be released by the Department of Medical Education and Research (DMER) Haryana after the national MCC MDS counselling process commences.
You can check, find and access more information here:
https://medicine.careers360.com/articles/haryana-neet-mds-counselling
Hi!
Given below is the link to access the NEET 2026 Physics Question Paper:
https://medicine.careers360.com/articles/neet-physics-question-paper
You can check the link below, apply relevant filters, find suitable resources related to NEET that will boost your overall preparation:
https://www.careers360.com/download/neet-sample-papers-previous-years
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs