JSS University Mysore Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Understanding the most frequently asked concepts in NEET Chemistry is key to smart preparation for NEET 2026. By analysing NEET Chemistry previous year questions (PYQs) from 2021 to 2025, aspirants can identify high-weightage chapters, repeated questions, and important topics that are consistently asked in the exam. This helps in focusing on scoring areas instead of covering the entire syllabus randomly.
This Story also Contains

NEET Chemistry questions are largely based on NCERT concepts from Physical, Organic, and Inorganic Chemistry. Topics like chemical bonding, thermodynamics, hydrocarbons, and coordination compounds are frequently asked. Regular revision of these high-yield topics, along with practising NEET PYQs with solutions, improves accuracy and boosts overall NEET exam performance.
This article provides a clear analysis of NEET Chemistry important concepts (2021–2025), including chapter-wise weightage, PYQ trends, and key topics to focus on for NEET 2026.
Let's understand the overall pattern of NEET Chemistry (2021–2025). The Chemistry section tests both conceptual clarity and application, with questions from physical, organic, and inorganic chemistry. Here’s a quick look at the question distribution, marks, and difficulty trends observed over the last five years:
Organic Chemistry questions have slightly increased since 2022, showing a stronger focus on reaction mechanisms and practical-based concepts. Studying NEET organic chemistry reactions is important for a good score.
Physical Chemistry remains calculation-based but often conceptual, testing the mole concepts, thermodynamics, and chemical kinetics.
Inorganic Chemistry questions are mostly fact-based and memory-driven, often repeated from previous years.
The overall difficulty level has remained moderate, with most questions derived directly from NCERT textbooks.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Based on the analysis of NEET Chemistry papers from 2021 to 2025, certain chapters and concepts are repeatedly tested. Focusing on these high-frequency topics can help students maximise their scores. Students can also check the top 5 NEET chemistry chapters for better preparation. Here’s a list of the most important NEET 2026 chemistry concepts:
Basic Concepts of Chemistry: Stoichiometry, limiting reagent, and empirical formula questions appear every year.
Thermodynamics: Enthalpy, Hess’s law, and Gibbs' free energy are frequently tested.
Chemical Kinetics: Rate laws, order of reaction, and half-life calculations.
Equilibrium: Le Chatelier’s principle, equilibrium constants, ionic equilibria.
Alcohols, Phenols, and Ethers: Preparation, properties, and reactions.
Aldehydes, Ketones and Carboxylic Acids: Nucleophilic addition reactions, oxidation, reduction.
Amines: Basicity, preparation, reactions with nitrous acid.
Hydrocarbons: Alkanes, alkenes, alkynes - addition, substitution, combustion reactions.
Chemical Bonding and Molecular Structure: VSEPR, hybridisation, shapes, polarity.
Coordination Compounds: Ligands, bonding, nomenclature, and isomerism.
d and f Block Elements: Transition metals, complexes, colour, and magnetic properties.
Understanding the chapter-wise weightage helps students focus on topics that carry the most marks and appear frequently in NEET Chemistry from 2021 to 2025.
The data is from the past five years, and chapters States of Matter, Hydrogen, The s-Block Element, Solid State, Surface Chemistry, Polymers, Environmental Chemistry, General Principles and Processes of Isolation of Elements are not in the NEET 2026 syllabus now
|
Chapter |
2021 |
2022 |
2023 |
2024 |
2025 |
Total weightage (%) |
|
1 |
1 |
1 |
1 |
3 |
3.91 | |
|
3 |
3 |
2 |
3 |
2 |
5.12 | |
|
Classification of Elements & Periodicity |
0 |
0 |
4 |
3 |
4 |
5.57 |
|
4 |
3 |
3 |
3 |
2 |
6.12 | |
|
States of Matter |
3 |
3 |
2 |
0 |
0 |
3.34 |
|
3 |
2 |
1 |
4 |
3 |
5.15 | |
|
1 |
1 |
1 |
3 |
1 |
2.97 | |
|
1 |
1 |
1 |
1 |
1 |
2.82 | |
|
Hydrogen |
0 |
1 |
1 |
0 |
0 |
0.84 |
|
The s-Block Element |
2 |
1 |
1 |
0 |
0 |
1.25 |
|
3 |
4 |
2 |
2 |
0 |
3.58 | |
|
2 |
3 |
2 |
1 |
1 |
3.43 | |
|
2 |
1 |
2 |
6 |
5 |
6.94 | |
|
Solid State |
2 |
2 |
2 |
0 |
0 |
2.34 |
|
3 |
3 |
0 |
5 |
3 |
6.33 | |
|
1 |
2 |
2 |
1 |
0 |
2.32 | |
|
1 |
2 |
1 |
2 |
3 |
4.73 | |
|
Surface Chemistry |
0 |
0 |
2 |
0 |
0 |
0.87 |
|
0 |
2 |
3 |
4 |
4 |
5.44 | |
|
3 |
3 |
1 |
5 |
4 |
6.63 | |
|
3 |
2 |
1 |
2 |
2 |
4.28 | |
|
2 |
2 |
4 |
2 |
1 |
4.28 | |
|
5 |
2 |
3 |
0 |
1 |
4.7 | |
|
Amines |
2 |
2 |
4 |
0 |
3 |
3.66 |
|
1 |
0 |
1 |
2 |
2 |
2.6 | |
|
Polymers |
1 |
1 |
1 |
0 |
0 |
1.25 |
|
Environmental Chemistry |
0 |
1 |
1 |
0 |
0 |
0.87 |
|
General Principles and Processes of Isolation of Elements |
1 |
2 |
1 |
0 |
0 |
1.28 |
This section provides previous year NEET Chemistry questions (2021–2025) based on the most frequently asked concepts. Students can use these important NEET chemistry questions to practice high-yield topics, understand repeated patterns, and improve their accuracy in the Chemistry section.
Ques: For a reaction
Chapter: Chemical Kinetics
Option 1)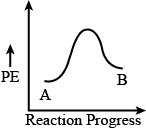
Option 2)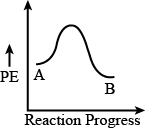
Option 3)
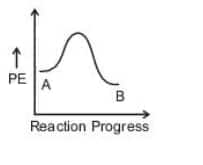
Option 4)
Correct answer: Option 2
Explanation: For an exothermic reaction, the enthalpy of the reactant will be greater than the enthalpy of the product and as a result, the energy profile diagram will look like
Hence, the answer is option (2).
Ques: Which one of the following alcohols reacts instantaneously with Lucas reagent? (NEET 2024)
Chapter: Alcohols Phenols and Ethers
Option 1)
Option 2)
Option 3)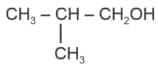
Option 4)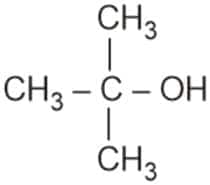
Correct answer: Option 4
Explanation: Tertiary alcohols react instantaneously with Lucas reagent and give immediate turbidity. In case of tertiary alcohols, they form halides easily with Lucas reagent.
Hence, the answer is option (4).
Ques: Given below are two statements:
Statement I: A hypothetical diatomic molecule with bond order zero is quite stable. Statement II: As the bond order increases, the bond length increases.
In light of the above statements, choose the most appropriate answer from the options given below:
Chapter: Chemical Bonding and Molecular Structure
Option 1) Statement 1 is false but Statement II is true
Option 2) Both Statement I and Statement II are true
Option 3) Both Statement I and Statement II are false
Option 4) Statement 1 is true but Statement II is false
Correct answer: Option 3
Explanation: A bond order of zero means no net bonding between the atoms, indicating the molecule cannot exist or is highly unstable. So the first statement is false
As the bond order increases, the bond length decreases because stronger bonding pulls the atoms closer together. And also the second statement is false
So the correct answer is (3) Both Statement I and Statement II are false
Hence, the correct answer is option (3).
Frequently Asked Questions (FAQs)
The basic concepts of chemistry for NEET include atomic structure, mole concept, chemical equations, stoichiometry, laws of chemical combination, concentration units, and dimensional analysis, which form the foundation for understanding physical, organic, and inorganic chemistry.
The hardest chapter in NEET Chemistry is often considered to be Thermodynamics or Organic Chemistry because they involve complex concepts and multiple formulas and require a deep understanding of theory as well as strong problem-solving skills.
The NEET 2025 Chemistry section was of moderate difficulty. Most questions were NCERT-based, but some required good conceptual understanding and application.
On Question asked by student community
Hello Dear Student,
First of all, don’t panic. Many students start serious NEET preparation in Class 12 and still crack the exam with a good score. What matters most now is consistency and smart planning, not when you started.
You should focus on these things:
Hello Dear Student,
With a NEET score of 520 and 73% in CBSE Class 12 , you may be eligible for:
in countries like:
However, fully funded scholarships usually require very high academic scores and stronger competitive
Hello Dear Student,
During NEET UG registration , candidates must enter the PCB aggregate percentage (Physics, Chemistry, Biology) and not the “Best of 5” percentage.
In your case, entering 90.8% instead of 89.2% is likely a calculation mistake rather than an eligibility issue, since both are above the required cutoff
Hi Akram!
Given below are the links to access NEET Previous Year Question Papers:
https://medicine.careers360.com/articles/neet-previous-year-question-paper-with-solution
https://medicine.careers360.com/articles/neet-previous-5-years-question-papers-with-solutions
Hello Sonam,
You may download the last 10 years of NEET question papers with solutions from the link below:
https://medicine.careers360.com/articles/neet-previous-year-question-paper-with-solution
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs