JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Chemical Kinetics NEET Previous Year Questions (2021–2025) analysis shows its high-weightage importance for NEET 2026. This chapter from NCERT Class 12 Chemistry consistently asks about concepts like rate of reaction, order of reaction, integrated rate equations, and half‑life calculations. Chemical Kinetics NEET questions are often numerical and formula‑based, making them most scoring for students who revise NCERT-based concepts thoroughly and important chemistry formulas. Solving these NEET PYQ provides knowledge about the mechanisms and factors affecting reactions.
This Story also Contains

Practising Chemical Kinetics NEET PYQs helps students identify repeated patterns, strengthen conceptual clarity, and improve problem‑solving speed. Since this chapter is included in the important topics of NEET 2026 Do or Die chapters, which include rate law, Arrhenius equation, and graphical interpretation of reaction kinetics are frequently asked questions. Clearing the NEET exam requires a smart and strategic approach as the syllabus is vast.
Chemical Kinetics helps aspirants learn about the Arrhenius equation and the collision theory of reactions. NEET chemistry chapter-wise weightage shows Chemical Kinetics holds 4.73% weightage. It is an important chapter of the NEET chemistry syllabus. Attempting NEET PYQ chemical kinetics helps students identify important topics. The table below shows the number of questions that appeared in the previous years.
|
Year |
No. of Questions |
|
2025 |
3 |
|
2024 |
2 |
|
2023 |
1 |
|
2022 |
2 |
|
2021 |
1 |
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Numerical-based questions from Chemical Kinetics appear in the NEET exam. To solve them in less time, an effective NEET preparation timetable is important. The timetable should include 2-3 hours daily for solving the NEET question paper. Provided below are a few NEET chemical kinetics previous year questions with solutions. Going through them makes students familiar with the question pattern
Question 1: If the half-life $(t_{1/2})$ for a first-order reaction is $1\ \text{minute}$, then the time required for $99.9%$ completion of the reaction is closest to: (NEET 2025)
Options
1. $10\ \text{minutes}$
2. $2\ \text{minutes}$
3. $4\ \text{minutes}$
4. $5\ \text{minutes}$
Answer: For a first-order reaction:
$t = \dfrac{6.9}{k}$ for $\sim 99.9%$ completion (more accurately, $t = \dfrac{6.91}{k}$)
Also, half-life is given by:
$t_{1/2} = \dfrac{0.693}{k} \Rightarrow k = \dfrac{0.693}{1} = 0.693$
$\Rightarrow t = \dfrac{6.91}{0.693} \approx 10\ \text{min}$
Hence, the correct answer is option $1)$ $10\ \text{minutes}$.
Question 2: If the rate constant of a reaction is $0.03\ \text{s}^{-1}$, how much time does it take for $7.2\ \text{mol L}^{-1}$ concentration of the reactant to get reduced to $0.9\ \text{mol L}^{-1}$? (Given: $\log 2 = 0.3$) (NEET 2025)
Options
1. $21.0\ \text{s}$
2. $69.3\ \text{s}$
3. $23.1\ \text{s}$
4. $210\ \text{s}$
Answer: Given that
Initial concentration $[R]_0 = 7.2\ \text{mol L}^{-1}$
Final concentration $[R] = 0.9\ \text{mol L}^{-1}$
Rate constant $k = 0.03\ \text{s}^{-1}$
Since units of $k$ are $\text{s}^{-1}$, it is a first-order reaction.
Formula:
$t = \dfrac{2.303}{k} \log \left( \dfrac{[R]_0}{[R]} \right)$
Substitute:
$t = \dfrac{2.303}{0.03} \log \left( \dfrac{7.2}{0.9} \right)$
$t = \dfrac{2.303}{0.03} \log (8)$
Now,
$\log(8) = \log(2^3) = 3 \log 2 = 3 \times 0.3 = 0.9$
So,
$t = \dfrac{2.303}{0.03} \times 0.9 = 76.77 \times 0.9 = 69.093\ \text{s}$
$\approx 69.3\ \text{s}$
Hence, the correct answer is option $2)$ $69.3\ \text{s}$.
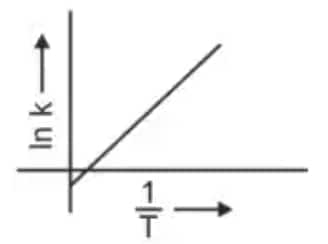
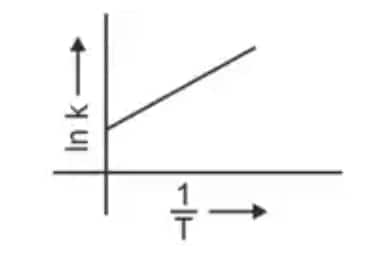
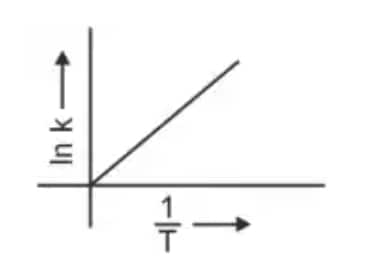
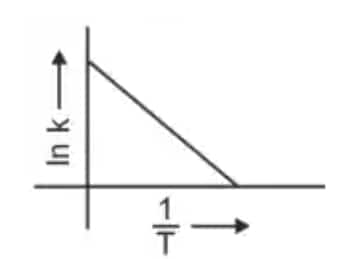
Question 3: Which plot of In k vs 1/T is consistent with the Arrhenius equation? (NEET 2024)
Options
3. 
4. 
Answer:
The Arrhenius equation is given as
$k = A e^{-\dfrac{E_a}{RT}}$
Taking logarithm
$\ln k = \ln A - \dfrac{E_a}{R}\left(\dfrac{1}{T}\right)$
Thus a plot of $\ln k$ vs $\dfrac{1}{T}$ gives a straight line
slope $= -\dfrac{E_a}{R}$
intercept $= \ln A$

Hence, the correct answer is option (4)
Question 4: For a certain reaction, the rate $R = k[A]^2[B]$, when the initial concentration of $A$ is tripled, keeping the concentration of $B$ constant, the initial rate would be: (NEET 2023)
Options
1. decrease by a factor of nine
2. increase by a factor of six
3. increase by a factor of nine
4. increase by a factor of three
Answer:
$R = k[A]^2[B]$
$r \propto [A]^2$, if $[B]$ remains constant
$r_f \propto [3A]^2 = 9[A]^2$
$\Rightarrow r_f \propto 9r_i$
So, the rate will increase $9$ times the initial rate.
Hence, the correct answer is option $(3)$, increase by a factor of nine.
Question 5: Given below are two statements: one is labeled as Assertion A and the other is labeled as Reason R:
Assertion A: A reaction can have zero activation energy.
Reason R: The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to the threshold value is called activation energy.
In light of the above statements, choose the correct answer from the options given below: (NEET 2023)
Options
1. Both A and R are true, and R is the correct explanation of A.
2. Both A and R are true, and R is NOT the correct explanation of A.
3. A is true, but R is false.
4. A is false, but R is true.
Answer:
Free radical reactions can have zero activation energy. In termination steps of free radical reactions, $E_a = 0$.
Hence, the correct answer is option $(2)$, Both A and R are true, and R is NOT the correct explanation of A.
Question 6: For a first-order reaction $A \rightarrow \text{Products}$, the initial concentration of $A$ is $0.1\ \text{M}$, which becomes $0.001\ \text{M}$ after $5$ minutes. Rate constant for the reaction in $\text{min}^{-1}$ is: (NEET 2022)
Options
1. $0.9212$
2. $0.4606$
3. $0.2303$
4. $1.3828$
Answer:
For a first-order reaction,
$\ln\left(\dfrac{[A]_0}{[A]_t}\right) = kt$
$\Rightarrow \ln\left(\dfrac{0.1}{0.001}\right) = k(5)$
$\Rightarrow k = \dfrac{\ln(100)}{5} = \dfrac{4.606}{5} = 0.9212\ \text{min}^{-1}$
Hence, the correct answer is option $(1)$, $0.9212$.
Question 7: The given graph is a representation of the kinetics of a reaction
The $y$ and $x$ axes for zero and first-order reactions respectively are: (NEET 2022)
Options
1. zero order $(y=\text{concentration}\ x=\text{time})$ first order $(y=\text{rate constant}\ x=\text{concentration})$
2. zero order $(y=\text{rate}\ x=\text{concentration})$ first order $(y=t_{1/2}\ x=\text{concentration})$
3. zero order $(y=\text{rate}\ x=\text{concentration})$ first order $(y=\text{rate}\ x=t_{1/2})$
4. zero order $(y=\text{concentration}\ x=\text{time})$ first order $(y=t_{1/2}\ x=\text{concentration})$
Answer:
For zero order reaction
$\text{Rate} \propto [A]^0$
$\Rightarrow \text{Rate} = k$
For first order reaction
$t_{1/2} = \dfrac{\ln 2}{k}$
Hence correct answer option $(2)$
Question 8: The slope of Arrhenius plot $\ln k$ vs $\dfrac{1}{T}$ for a first-order reaction is $-5 \times 10^3\ \text{K}$. The value of $E_a$ is: (NEET 2021)
Given $R = 8.314\ \text{J K}^{-1}\text{mol}^{-1}$
Options
1. $41.5\ \text{kJ mol}^{-1}$
2. $83.0\ \text{kJ mol}^{-1}$
3. $166\ \text{kJ mol}^{-1}$
4. $-83\ \text{kJ mol}^{-1}$
Answer:
$\ln k = \ln A - \dfrac{E_a}{R}\left(\dfrac{1}{T}\right)$
Slope $m = -\dfrac{E_a}{R}$
$-5 \times 10^3 = -\dfrac{E_a}{8.314}$
$\Rightarrow E_a = 5 \times 10^3 \times 8.314$
$E_a = 41.5\ \text{kJ mol}^{-1}$
Hence correct answer option $(1)$
Question 9: An increase in the concentration of the reactants of a reaction leads to a change in: (NEET 2020)
Options
1. Collision frequency
2. Activation energy
3. Heat of reaction
4. Threshold energy
Answer:
Increase in concentration increases number of collisions
Hence correct answer option $(1)$
Question 10: The rate constant for a first-order reaction is $4.606 \times 10^{-3}\ \text{s}^{-1}$. The time required to reduce $2\ \text{g}$ of reactant to $0.2\ \text{g}$ is: (NEET 2020)
Options
1. $1000\ \text{s}$
2. $100\ \text{s}$
3. $200\ \text{s}$
4. $500\ \text{s}$
Answer:
$kt = \ln\left(\dfrac{A_0}{A_t}\right)$
$4.606 \times 10^{-3} t = 2.303 \log\left(\dfrac{2}{0.2}\right)$
$= 2.303$
$\Rightarrow t = \dfrac{2.303}{4.606 \times 10^{-3}}$
$t = 500\ \text{s}$
Hence correct answer option $(4)$
Chemical Kinetics is a scoring unit in chemistry. It is a high-weightage chemistry chapter with consistently asked questions in previous years. Chemical kinetics NEET PYQ includes questions on how reactions occur and the different factors affecting them. Students thinking about how to study chemistry for NEET should start with this unit. Given below are a few important topics from this unit
|
Topics |
Description |
|
Defined as the change in concentration of reactants or products per unit time. | |
|
Order of Reaction |
For complex reactions, the order is determined by the rate-determining step. |
|
The number of reacting species involved in an elementary reaction. | |
|
Rate Constant (k) |
A proportionality constant that relates the rate of the reaction to the concentrations of the reactants. |
|
Activation Energy (Eₐ) |
The minimum energy required for a reaction to occur. |
|
This equation relates the rate constant to temperature and activation energy. It is important for understanding how temperature affects reaction rates. | |
|
Half-Life (t₁/₂) |
The time required for half of the reactant to be consumed is known as half half-life. |
|
Factors Affecting Reaction Rates |
Understanding how concentration, temperature, and catalysts influence reaction rates is important. |
Solving previous year NEET questions regularly builds confidence. It helps students understand the types of questions asked in the exam. Given below are a few tips that they can use to solve NEET chemical kinetics previous year questions.
Prepare flashcards of important formulas for NEET chemistry. This helps to answer NEET chemical kinetics previous year questions confidently.
Frequently Asked Questions (FAQs)
Because it is a Do or Die chapter with repeated PYQs on rate of reaction, order of reaction, integrated rate equations, and half‑life, making it highly scoring.
On average, 2–3 questions appear every year, mostly numerical and formula‑based, directly from NCERT Class 12 Chemistry.
Focus on rate law, Arrhenius equation, half‑life formulas, integrated rate equations, and graphical interpretation of reaction kinetics.
On Question asked by student community
Hello Dear Student,
No, NEET is not compulsory for Bachelor of Physiotherapy (BPT) admissions for the 2026–27 academic session.
You can check, find and access more information here:
https://news.careers360.com/physiotherapy-neet-admission-1yr-internship-bpt-dr-title-allied-health-sciences-paramedical-courses-revamp-syllabus-ncahp-mohfw
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-mds-2025-seat-allotment-result
Hope it helps!
Hello Dear Student,
With a NEET MDS 2026 score of 503 and an AIR of around 4600, getting a government seat in highly competitive branches such as Orthodontics, Conservative Dentistry & Endodontics, or Prosthodontics may be difficult, as these specialties usually close at much better ranks.
However, you still have
Hello Dear Student,
Haryana NEET MDS 2026 counselling registrations are expected to begin in July 2026. The official schedule will be released by the Department of Medical Education and Research (DMER) Haryana after the national MCC MDS counselling process commences.
You can check, find and access more information here:
https://medicine.careers360.com/articles/haryana-neet-mds-counselling
Hi!
Given below is the link to access the NEET 2026 Physics Question Paper:
https://medicine.careers360.com/articles/neet-physics-question-paper
You can check the link below, apply relevant filters, find suitable resources related to NEET that will boost your overall preparation:
https://www.careers360.com/download/neet-sample-papers-previous-years
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs