JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
If you want to become a doctor, it is important that you get a good rank in the National Eligibility cum Entrance Test (NEET) for undergraduate (UG) admissions to medical courses. And for this, you need to master all the subjects (Physics, Chemistry and Biology) included in the NEET UG syllabus. A good grip on Physics is crucial as 25% of the questions in the NEET paper is from the subject. So, how well you prepare for Physics is crucial in deciding your rank in the NEET exam, which is conducted annually.

Knowing the important topics from each chapter will be quite useful in your preparation. Here is an analysis of the previous five years' NEET questions from the Class 12 NCERT chapter, Dual Nature Of Matter And Radiation. In the Last five years of NEET paper, a total of 9 questions were asked from the Class 12 NCERT chapter, Dual Nature Of Matter And Radiation.
Topic | Number Of Questions |
Einstein’s Photoelectric Equation | 3 |
de-Broglie Wavelength | 3 |
Concept of Electron Volt | 1 |
Energy of Photon | 1 |
Intensity of Light | 1 |
The above table makes it clear that Einstein's Photoelectric Equation and the de-Broglie Wavelength are two important topics of the chapter, Dual Nature Of Matter And Radiation. Let us get a brief idea of these concepts and the NEET questions related to them.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
The photoelectric equation is given by -
Kinetic Energy (K.E) of the photo-electrons = (Energy obtained from the Photon) – (The energy used to escape the metallic surface).
KE=h?-ɸ
ɸ=h?0
The energy used to escape the metallic surface is known as the work function (ɸ). Here ?0 is the threshold frequency.
The photoelectric threshold wavelength of silver is 3250 x 10-10 m. The velocity of the electron ejected from a silver surface by the ultraviolet light of wavelength 2536 x 10-10 m is:
(Given h = 4.14 x 10-15 eVs and c = 3 x 108 ms-1)
Solution
Here the wavelengths are given.
![]()
Therefore the photoelectric equation can be written as
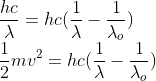
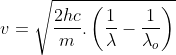
Given
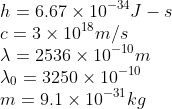
Substituting the values the answer is
![]()
When the light of frequency 2?0 (where ?0 is threshold frequency), is incident on a metal plate, the maximum velocity of electrons emitted is ?1. When the frequency of the incident radiation is increased to 5?0, the maximum velocity of electrons emitted from the same plate is ?2. The ratio of ?1 to ?2 is
Solution
From the Einstein equation
![]()
Initially
![]()
Finally
![]()
![]() or
or
![]()
An electromagnetic wave of wavelength λ is incident on a photosensitive surface of negligible work function. If 'm' mass is of photoelectron emitted from the surface has de-Broglie wavelength λd then:
Solution
![]()
Momentum P = h/λd where λd is the de-Broglie wavelength, work function ɸ=0(Given)
![]()
![]()
![]()
The de-Broglie wavelength of electron λe is given by
![]()
![]()
![]()
![]()
![]()
The de Broglie Wavelength Associated With Charged Particle
![]()
Where K is the Kinetic Energy, m is the mass of the particle, q is the charge of the particle and V is the potential difference.
Recommended Video-
The de-Broglie wavelength of a neutron in thermal equilibrium with heavy water at a temperature T (Kelvin) and mass m, is
Solution
Kinetic energy of the neutron in thermal equilibrium with heavy water is-
![]()
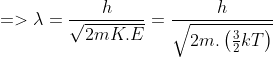
Here K.E is the Kinetic Energy
![]()
An electron of mass m with an initial velocity
![]()
enters an electric field
![]()
at t = 0 . If ![]() is its de-Broglie wavelength initially, then its de-Broglie wavelength at time t is
is its de-Broglie wavelength initially, then its de-Broglie wavelength at time t is
Options
![]()
![]()
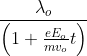
![]()
Solution
![]()
![]()
![]()
At time t the magnitude of the velocity
![]()
Momentum
![]()
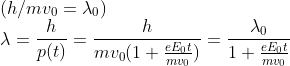
So option C is the right answer.
An electron is accelerated through a potential difference of 10000 V . Its de Broglie wavelength is
![]()
Solution
Use the equation-
![]()
![]()
In the last 5 years’ (2017-2021) NEET papers, 9 questions were asked and out of these 6 were from the topics de Broglie Wavelength and Einstein's Photoelectric Equation. That is, 66.67% of questions from Dual Nature were from these two topics.
On Question asked by student community
Hello Dear Student,
Yes, qualifying for the NEET is mandatory if you are seeking admission to core medical courses like
MBBS
,
BDS
,
BAMS
,
BHMS
, and
BSc Nursing
across India.
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-eligibility-criteria
Hope it helps!
Hello Dear Student,
If you are a PCB student and want to keep your career options open apart from NEET, there are several excellent courses and entrance exams you can consider.
Agriculture & Allied Sciences
ICAR UG admissions for courses such as B.Sc. Agriculture, Horticulture, Forestry, Fisheries, and related fields.
Hello Dear Student,
The NEET syllabus can seem overwhelming at first, but with a proper strategy and consistent effort, it becomes much more manageable. The key is to focus on the most important topics and practice regularly.
Effective NEET Preparation Tips
Prioritize NCERT Books: NCERT should be your primary study
Hello Dear Student,
You generally do not need to upload your physical category certificate during the initial online application. However, you must declare your category (OBC-NCL) to claim reservation benefits and enter the certificate details. You must provide the physical certificate during counseling and document verification.
You can check, find
Hello Dear Student,
For the NEET PG application form, you do not need to upload your actual OBC or OBC-NCL (Non-Creamy Layer) certificate. You only need to declare your category and claim the OBC-NCL reservation during the registration phase.
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-caste-certificate-2025
Hope
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs