JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Important NEET 2026 Chemistry Questions are a must-practice resource for aspirants aiming to secure high marks in the Chemistry section. Analysis of NEET Chemistry PYQs (2021-2025) shows that questions are repeatedly asked from the Physical, Organic, and Inorganic Chemistry chapters. Focusing on high-weightage NEET Chemistry questions, frequently asked concepts, and previous year trends can significantly improve accuracy and speed. Many aspirants actively search for important NEET Chemistry questions, chapter-wise practice MCQs, and quick revision resources to improve their NEET 2026 preparation in the final phase.
This Story also Contains
.jpg)
Practising the most important NEET Chemistry questions helps students strengthen conceptual clarity, improve calculation speed, and avoid common mistakes under exam pressure. To make preparation easier, we have compiled the Top 20 Important NEET Chemistry Questions based on previous years’ patterns. These questions cover the most important and frequently asked concepts. For a higher score, students can also follow a proven strategy on how to score 160+ in NEET Chemistry. Regular practice and revision will boost confidence and overall performance in the NEET exam.
Get the NEET 2026 Chemistry important questions PDF with the most scoring NEET concepts and problems from Physical, Organic, and Inorganic Chemistry. This compact resource includes solved PYQs, formula‑based numericals, and conceptual MCQs for quick revision. Download the PDF to strengthen last-minute preparation and boost your Chemistry score.
|
NEET Chemistry Top 20 Important Questions PDF |
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
This part is a combination of concept, numerical, and application-type questions of Physical, Organic, and Inorganic Chemistry. These are the most commonly asked questions. This will not only improve your NEET 2026 last-minute preparation but also improve speed and accuracy during the exam. We have presented here the most relevant NEET Chemistry questions along with detailed answers so that you can approach the exam questions confidently and effectively.
Ques:
Given below are two statements :
Statement I : A hypothetical diatomic molecule with bond order zero is quite stable. Statement II : As bond order increases, the bond length increases.
In the light of the above statements, choose the most appropriate answer from the options given below :
Option 1) Statement 1 is false but Statement II is true
Option 2) Both Statement I and Statement II are true
Option 3) Both Statement I and Statement II are false
Option 4) Statement 1 is true but Statement II is false
Chapter - Chemical bonding and molecular structure
Difficulty level - Easy
Solution -
A bond order of zero means no net bonding between the atoms, indicating the molecule cannot exist or is highly unstable. so the first statement is false
As bond order increases, bond length decreases because stronger bonding pulls the atoms closer together. and also the second statement is false
So the correct answer is (3) Both Statement I and Statement II are false
Hence, the correct answer is option (3).
Ques:
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : 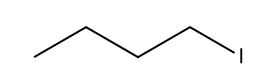
undergoes SN2 reaction faster than
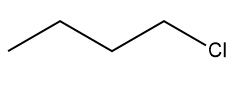
Reason ( RR ) : lodine is a better leaving group because of its large size.
In the light of the above statements, choose the correct answer from the options given below :
Option 1) A is false but R is true
Option 2) Both A and R are true and R is the correct explanation of A
Option 3) Both A and R are true but R is not the correct explanation of A
Option 4) A is true but R is false
Chapter - Haloalkanes and Haloarenes
Difficulty level - Medium
Solution -
Rate α [RX][Z-]
where Z- is the leaving group
Since iodide is a better leaving group than chloride so the SN2 reaction will be faster in the iodobutane.
The larger size of iodine results in a weaker bond between iodine and the carbon atom it's attached to that makes it easier to dissociate and leave during a reaction.
Hence, the correct answer is option (2).
Ques:
C( s)+2H2( g)→CH4( g);ΔH=−74.8 kJ mol−1
Which of the following diagrams gives an accurate representation of the above reaction? [R→ reactants; P→ products ]
Option 1) 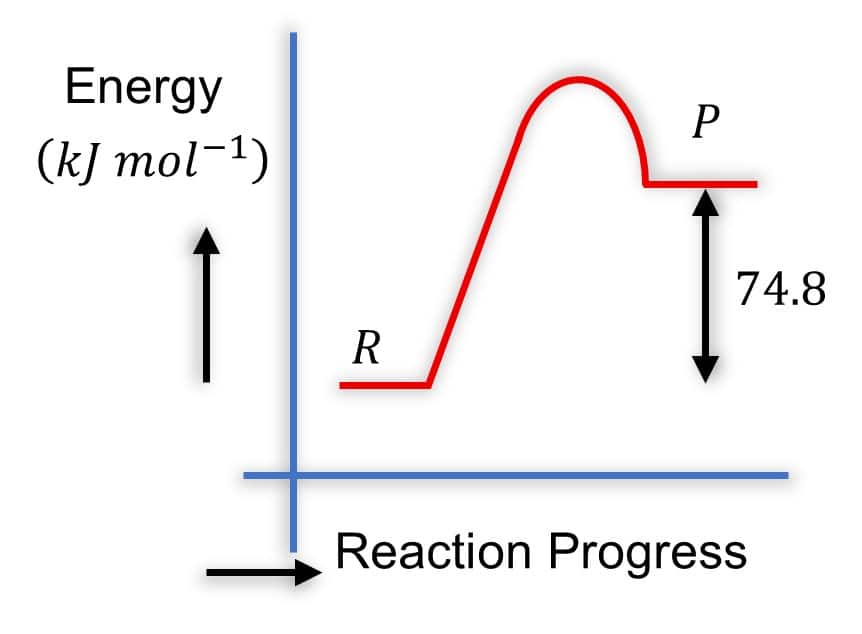
Option 2) 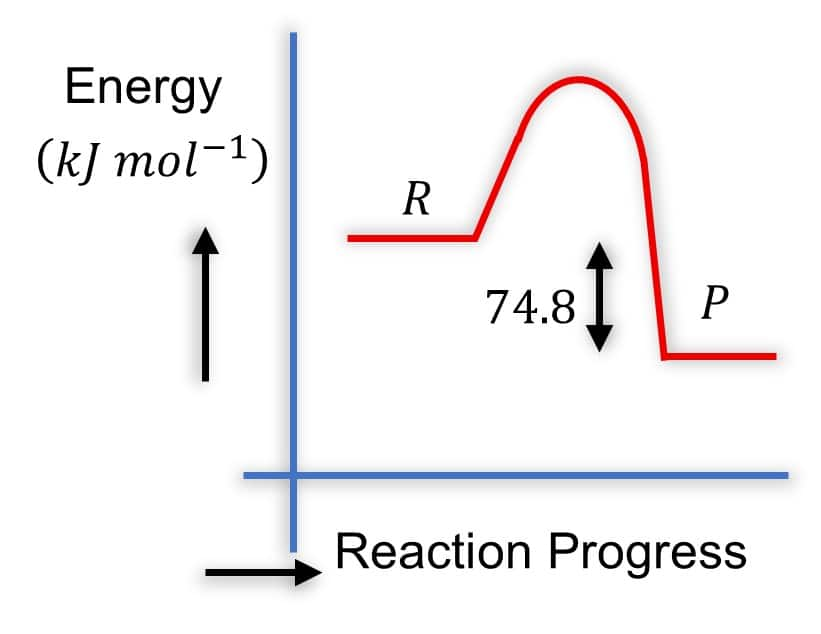
Option 3) 
Option 4) 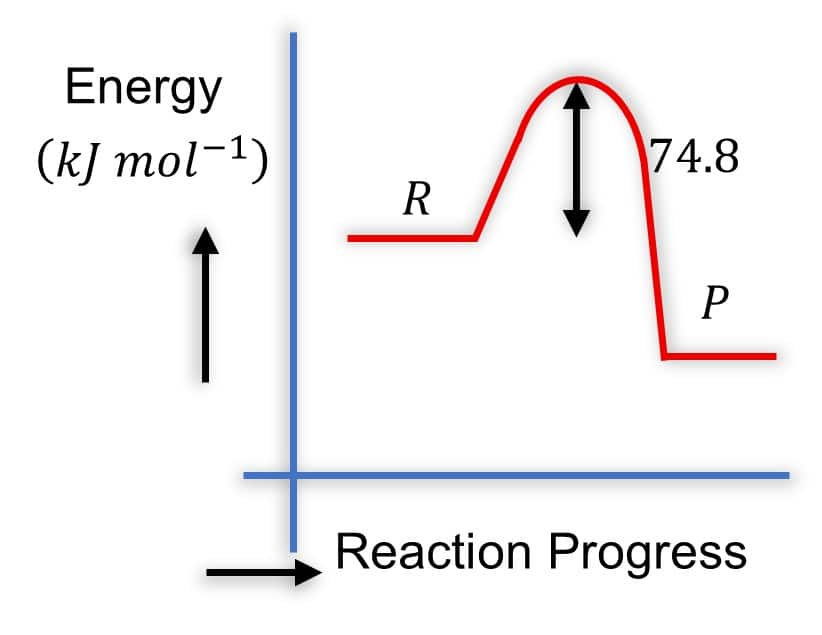
Chapter - Thermodynamics
Difficulty Level - Medium
Solution - Diagram 2:
- R is higher than P
- P is lower in energy
- The arrow from P to R is labeled 74.8 and points upward
- This is correct because it represents the energy difference, and ΔH=−74.8kJmol−1.
The arrow shows that products are lower by 74.8kJmol−1.
- It is common to show the magnitude of ΔH with an upward arrow between P and R even in exothermic reactions, as long as positions are correct.
Hence, the correct answer is option (2).
The chemistry NEET 2026 has all three parts, which are Physical, Organic, and Inorganic Chemistry from Class 11 and Class 12. The table below shows NEET chemistry chapter-wise weightage for the last 5 years. Aspirants will also get to know the difficulty level of NEET chemistry important questions.
The data is from the last five years and chapters States of Matter, Hydrogen, The s-Block Element, Solid State, Surface Chemistry, Polymers, Environmental Chemistry, General Principles and Processes of Isolation of Elements are not in the NEET syllabus now
| Chapter | 2021 | 2022 | 2023 | 2024 | 2025 |
Total weightage (%) |
| Basic Concepts of Chemistry | 1 | 1 | 1 | 1 | 3 | 3.91 |
| Structure of Atom | 3 | 3 | 2 | 3 | 2 | 5.12 |
| Classification of Elements & Periodicity | 0 | 0 | 4 | 3 | 4 | 5.57 |
| Chemical Bonding and Molecular Structure | 4 | 3 | 3 | 3 | 2 | 6.12 |
| States of Matter | 3 | 3 | 2 | 0 | 0 | 3.34 |
| Thermodynamics | 3 | 2 | 1 | 4 | 3 | 5.15 |
| Equilibrium | 1 | 1 | 1 | 3 | 1 | 2.97 |
| Redox Reactions | 1 | 1 | 1 | 1 | 1 | 2.82 |
| Hydrogen | 0 | 1 | 1 | 0 | 0 | 0.84 |
| The s-Block Element | 2 | 1 | 1 | 0 | 0 | 1.25 |
| Some p-Block Elements | 3 | 4 | 2 | 2 | 0 | 3.58 |
| d and f Block Elements | 2 | 3 | 2 | 1 | 1 | 3.43 |
| Coordination Compounds | 2 | 1 | 2 | 6 | 5 | 6.94 |
| Solid State | 2 | 2 | 2 | 0 | 0 | 2.34 |
| Solutions | 3 | 3 | 0 | 5 | 3 | 6.33 |
| Electrochemistry | 1 | 2 | 2 | 1 | 0 | 2.32 |
| Chemical Kinetics | 1 | 2 | 1 | 2 | 3 | 4.73 |
| Surface Chemistry | 0 | 0 | 2 | 0 | 0 | 0.87 |
| Organic Chemistry – Some Basic Principles and Techniques | 0 | 2 | 3 | 4 | 4 | 5.44 |
| Hydrocarbons | 3 | 3 | 1 | 5 | 4 | 6.63 |
| Haloalkanes and Haloarenes | 3 | 2 | 1 | 2 | 2 | 4.28 |
| Alcohols, Phenols and Ethers | 2 | 2 | 4 | 2 | 1 | 4.28 |
| Aldehydes, Ketones and Carboxylic Acids | 5 | 2 | 3 | 0 | 1 | 4.7 |
| Amines | 2 | 2 | 4 | 0 | 3 | 3.66 |
| Biomolecules | 1 | 0 | 1 | 2 | 2 | 2.6 |
| Polymers | 1 | 1 | 1 | 0 | 0 | 1.25 |
| Environmental Chemistry | 0 | 1 | 1 | 0 | 0 | 0.87 |
| General Principles and Processes of Isolation of Elements | 1 | 2 | 1 | 0 | 0 | 1.28 |
Frequently Asked Questions (FAQs)
The most important Chemistry questions for NEET 2026 are based on high-weightage chapters like Chemical Bonding, Coordination Compounds, Hydrocarbons, Thermodynamics, and Solutions, as these topics are frequently asked in previous years.
Students should practice NCERT-based questions, previous year questions (PYQs), and mock tests regularly. Revising formulas, reactions, and solving topic-wise MCQs helps improve accuracy and speed.
Yes, NCERT is the most important resource for NEET Chemistry, especially for the Inorganic and Organic sections. However, students should also practice PYQs and application-based questions to strengthen concepts and perform well in the exam.
On Question asked by student community
Hello Dear Student,
With a NEET MDS 2026 score of 225 , getting MDS Prosthodontics in a government dental college is likely to be difficult, especially in the General category, as Prosthodontics is one of the more competitive MDS branches. Recent government college cutoffs for Prosthodontics have generally been much
Hello,
If you are not eligible for NEET MDS , admission to an MDS course is generally not possible until you meet the eligibility requirements. To appear for NEET MDS, candidates must have a recognized BDS degree, complete the mandatory internship, and have registration with the Dental Council.
Depending on
Hello Dear Student,
With a NEET MDS rank of 8279 and BCD category , getting an MDS government seat is likely to be difficult, especially in high-demand clinical branches such as Orthodontics, Prosthodontics, Oral Surgery, and Conservative Dentistry. Recent government college cutoffs for OBC-category candidates in these branches have generally
Hello,
As of now, the revised NEET MDS 2026 eligibility/qualifying score has not been officially announced by NBEMS . The current qualifying criteria remain:
General/EWS: 50th percentile
SC/ST/OBC: 40th percentile
General PwD: 45th percentile
Hello Dear Student,
With a NEET MDS score of 347 and AIR around 12,324 (General category) , your chances for top government dental colleges are quite low , as government MDS seats usually close at much better ranks, especially for clinical branches. Recent marks-vs-rank data places scores in the 334–449
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs