JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
NEET 2026 Mock Test Day 1 is now live (10 AM–5 PM)! Attempt this full-length paper in real exam conditions and analyse your performance with the same-day answer key and solutions. Based on the latest update shared by NTA on X (formerly Twitter), this mock test series is designed to align with current NEET 2026 preparation trends and expected exam patterns.
This Story also Contains

In the final phase of preparation, solving full-length mock tests, NCERT-based questions, and previous year papers is far more effective than passive revision. This test covers Physics, Chemistry, and Biology with real exam-level difficulty to help you improve accuracy, speed, and confidence in the NEET exam.
This NEET 2026 Mock Test Day 1 replicates the actual exam experience and helps you assess your preparation under real-time conditions. This structured approach is widely followed by top NEET rankers to improve accuracy and performance under pressure.
Full-length NEET mock test (180 questions)
Covers Physics, Chemistry, and Biology
Based on NCERT and previous year trends
Matches real NEET difficulty level
Includes same-day answer key and solutions
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
To get the maximum benefit from the NEET 2026 Mock Test Day 1, it’s important to follow a disciplined, exam-like approach rather than casual practice. This strategy helps improve accuracy, time management, and real exam performance.
Attempt the full paper before 10 AM to match the actual exam timing
Solve the test within 3 hours under strict exam conditions
Do not check answers or pause during the attempt
Review the answer key after 5 PM
Analyse mistakes thoroughly and revise weak areas on the same day
This structured routine creates a same-day improvement cycle, a proven method commonly followed by top NEET rankers to improve scores in the final days.
Instructions:
Q-1:-
A particle is moving such that its position coordinates (x,y) are (2m, 3m) at time t = 0, (6m, 7m) at time t = 2 s and (13m, 14m) at time t = 5s. Average velocity vector D from t = 0 to t = 5 s is:
Q-2:-
A person trying to lose weight by burning fat lifts a mass of 10 kg to a height of 1 m, 1000 times. Assume that the potential energy lost each time he lowers the mass is dissipated. How much fat will he use up considering the work done only when the weight is lifted up? Fat supplies 3.8×107 J of energy per kg which is converted to mechanical energy with a 20% efficiency rate. (Take g=9.8 ms−2)
1)
2.45×10−3 kg
2)
6.45×10−3 kg
3)
9.89×10−3 kg
4)
12.89×10−3 kg
Q-3:-
A nucleus of uranium decays at rest into nuclei of thorium and helium. Then:
1)
The helium nucleus has less momentum than the thorium nucleus.
2)
The helium nucleus has more momentum than the thorium nucleus.
3)
The helium nucleus has less kinetic energy than the thorium nucleus.
4)
The helium nucleus has more kinetic energy than the thorium nucleus.
Q-4: The acceleration of an electron in the first orbit of the hydrogen atom $(n = 1)$ is:
Q-5:-
In a physical balance working on the principle of moments, when 5 mg weight is placed on the left pan, the beam becomes horizontal. Both the empty pans of the balance are of equal mass. Which of the following statements is correct?
1)
Left arm is longer than the right arm
2)
Both the arms are of same length
3)
Left arm is shorter than the right arm
4)
Every object that is weighed using this balance appears lighter than its actual weight.
Q-6:-
A pendulum with time period of 1,s is losing energy due to damping. At a certain instant, its energy is 45,J. After completing 15 oscillations, its energy becomes 15,J. The damping constant (in s−1) is:
Q-7:-
$PV = nRT + \alpha V$,
where $n$ is the number of moles and $\alpha$ is a positive constant. The initial temperature and pressure of one mole of the gas are $T_0$ and $P_0$, respectively. The work done by the gas when its temperature doubles under isobaric conditions is:
Q-8:-
A black-coloured solid sphere of radius $R$ and mass $M$ is placed inside a cavity maintained at temperature $T_0$ (vacuum inside). The initial temperature of the sphere is $3T_0$. The specific heat of the material varies as $\alpha T^3$ per unit mass, where $\alpha$ is a constant. The time taken for the sphere to cool down to temperature $2T_0$ is (given $\sigma$ is the Stefan–Boltzmann constant):
Q-9:-
A parallel plate capacitor with air between the plates has a capacitance of $9,\text{pF}$. The separation between the plates is $d$. The space between the plates is now filled with two dielectrics: one with dielectric constant $k_1 = 3$ and thickness $\dfrac{d}{3}$, and the other with dielectric constant $k_2 = 6$ and thickness $\dfrac{2d}{3}$. The new capacitance of the capacitor is:
Q-10:-
Which of the following cannot be emitted by radioactive substances during their decay?
1)
protons
2)
neutrinos
3)
helium nuclei
4)
electrons
Q-11:-
The given graph represents V - I Characteristic for a semiconductor device.
Which of the following statement is correct?
1)
It is V - I Characteristic for solar cell where, point A represents open circuit voltage and point B Short circuit current.
2)
It is a for a solar cell, and point B represents open circuit voltage and current, respectively.
3)
It is a for a photodiode and poins A and B represent open circuit voltage and current respectively.
4)
It is a for a LED points And B represent open circuit voltage and short circuit current respectively.
Q-12:-
The equivalent capacitance between A and B in the circuit given below, is :

Q-13:-
In the given circuit all resistances are of value R ohm each. The equivalent resistance between A and B is :

1)
2R
2)
3R
3)
5R3
4)
5R2
Q-14:-
A solid cylinder of radius R carrying on current I. Find the magnetic field at a point P due to cylinder is P is at r>R
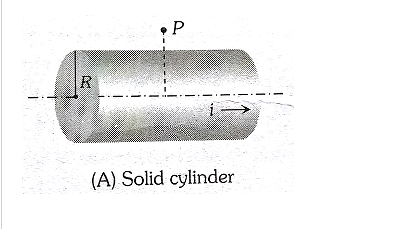
Q-15:-
The susceptance of a circuit is
1) 1R
2) 1Z
3) 1X
4) None
Q-16:-
A mirror of radius of curvature 20cm and an object which is placed at a distance of 15cm are both moving with velocity 1ms-1 and 10ms-1 . The velocity of image in this situation is
1) 40 cm/s
2) 44 cm/s
3) 45 cm/s
4) 30 cm/s
Q-17:-
The efficiency of a practical transformer is (where PL = power loss):
Q-18:-
A small mass m is slipping over a frictionless incline made of wood of mass M. The acceleration by which the incline should be pushed so that block m remains stationary w.r.t incline is

1)
23g
2)
g3
3)
g2
4)
32g
Q-19:-
If the coefficient of friction between an insect and a bowl is μ and the radius of the bowl is r, the maximum height to which the insect can crawl up the bowl is:
Q-20:-
Who among the following gave first the experimental value of G (Universal gravitational constant)?
1)
Brook Teylor
2)
Copernicus
3)
Cavendish
4)
None of these
Q-21:-
If 5,mg of a liquid at temperature T and specific heat 2s is mixed with 5,mg of another liquid at temperature 2T and specific heat 3s, the final temperature of the mixture is:
Q-22:-
For an adiabatic process the value of specific heat is
1)
Zero
2)
Infinitely
3)
finite and Positive
4)
A person standing on open ground hears the sound of a jet aeroplane coming from the north at an angle 60∘ with the ground. However, he observes the aeroplane to be vertically above him. If v is the speed of sound, the speed of the aeroplane is:
Q-24:-
A potentiometer wire AB having length L and resistance 12r is joined to a cell D of emf ε and internal resistance r. A cell Chaving emf ε2 and internal resistance 3 r is connected. The length AJ is connected with the galvanometer as shown in the fig. shows no deflection is:
1)
1112L
2)
1124L
3)
1324L
4)
512L
Q-25:-
An upright object is placed at a distance of 40 cm in front of a convergent lens of focal length 20 cm. A convergent mirror of focal length 10 cm is placed at a distance of 60 cm on the other side of the lens. The position and size of the final image will be :
1)
20 cm from the convergent mirror, same size as the object
2)
20 cm from the convergent mirror, twice the size of the object
3)
40 cm from the convergent mirror, same size as the object
4)
40 cm from the convergent lens, same size of the object
Q-26:-
What is the dimension of Mass?
1)
[M]
2)
[L]
3)
[T]
4)
[K]
Q-27:-
If the speed and mass of an object are doubled, which of the following is true?
1)
The momentum of the object is doubled
2)
The momentum of the object is quadrupled.
3)
The momentum remains same.
4)
The momentum is halved.
Q-28:-
Which of the following can be explained by the Huygens principle?
1)
Snell's law
2)
Laws of reflection
3)
Both A and B
4)
None of these
Q-29:-
A plane electromagnetic wave is propagating along the direction $(\hat{i} + 2\hat{j})$, with its electric field polarized along $\hat{k}$. The correct form of the magnetic field of the wave is (where $B_0$ is a constant):
Q-30:-
In Young's double-slit experiment, if the separation between coherent sources is halved and the distance of the screen from the coherent courses is doubled, the fringe width becomes -
1)
double
2)
half
3)
four times
4)
one-fourth
Q-31:-
The temperature of a room heated by a heater is $20^\circ\text{C}$ when the outside temperature is $-20^\circ\text{C}$, and it is $10^\circ\text{C}$ when the outside temperature is $-40^\circ\text{C}$. The temperature of the heater is:
Q-32:-
Laws of Electromagnetic induction were discovered by
1)
Chadwick
2)
Faraday
3)
Newton
4)
Einstein
Q-33:-
The weight of an astronaut in an orbiting artificial satellite around Earth is
1)
Zero
2)
Same as on Earth
3)
More than on Earth
4)
Less than on Earth
Q-34:-
Assertion: Balmer series lies in the visible region of the electromagnetic spectrum.
Reason:
1)
Both Assertion and Reason are correct and Reason is the correct explanation for Assertion
2)
Both Assertion and Reason are correct but Reason is not the correct explanation for Assertion
3)
Assertion is correct but Reason is incorrect
4)
Assertion is incorrect but Reason is correct
Q-35:-
A person walks up a stalled escalator in 90 sec. When standing on the same escalator, now moving, he is carried in 60 sec. The time it would take him to walk up the moving escalator will be
1)
27 sec.
2)
72 sec.
3)
18 sec.
4)
36 sec.
Q-36:-
The position of a particle moving along the $x$-axis is given by
$x = 2At^3 + 4Bt^2 + 40Ct + D$.
The numerical values of $A, B, C,$ and $D$ are $1, 4, -2,$ and $5$ respectively. The acceleration of the particle at $t = 1,\text{s}$ is:
Q-37. What is the charge on an α-particle?
Q-38:-
Characteristic X-rays of frequency $4.2 \times 10^{18},\text{Hz}$ are produced due to transitions to the K-shell in a target material. Using Moseley’s law, determine the atomic number of the target.
(Given: Rydberg constant $R = 1.1 \times 10^7,\text{m}^{-1}$)
1) Z = 32
2) Z = 42
3) Z = 41
4) Z = 44
Q-39:-
The RMS value of the electric field of sunlight is 720,N/C. The average total energy density of the electromagnetic wave is:
Q-40:-
Currents of 3A,1A and 2A flow through the long, Straight and parallel conductors a, b and c respectively as shown: A length 0.5m of wire b experiences a force if
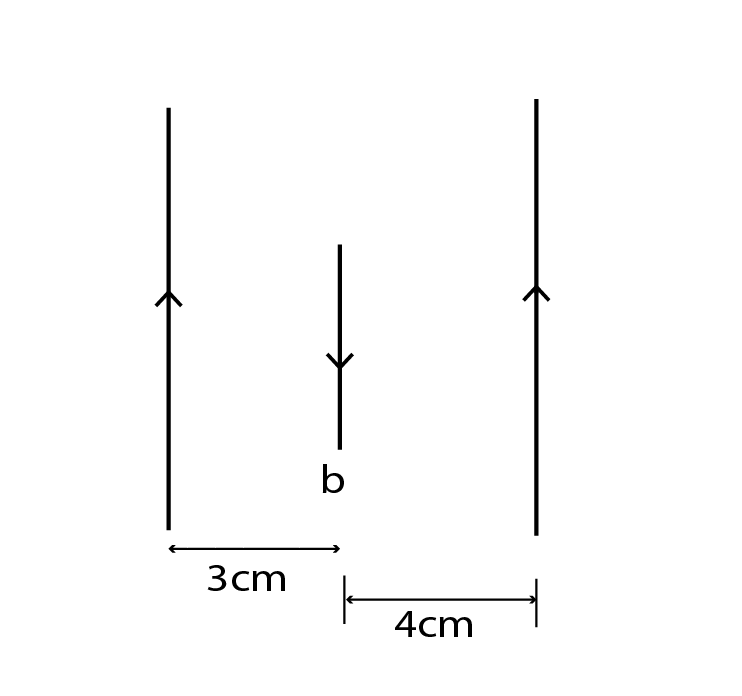
Q-41:-
A bar magnet of magnetic moment M→ is placed in a magnetic field of induction B→. The torque acting on it is:
Q-42:-
In a special experiment, angular momentum is an even integral multiple of h2π. The longest possible wavelength emitted by hydrogen in the visible region in the experiment according to Bohr’s model will be:
1) 478,nm
2) 322,nm
3) 32 nm
4) 50 nm
Q-43:-
Ten moles of an ideal gas at a constant temperature of $600,\text{K}$ is compressed from $100,\text{L}$ to $10,\text{L}$. The work done in the process is:
Q-44:-
A uniform spherical shell of mass 1 kg and radius 1 m is rolling with slipping on a level road. find the distance covered by the particle at the point of contact when the shell completes half revolution.
1)
R
2)
2R
3)
3R
4)
4R
Q-45:-
Two heaters A and B have power rating of 1 kW and 2 kW, respectively. Those two are first connected in series and then in parallel to a fixed power source. The ratio of power outputs for these two cases is:
1)
1:1
2)
2:9
3)
1:2
4)
2:3
Q-1:-
Which of the following lanthanoid ions is diamagnetic?
(At no.s. Ce = 58, Sm = 62, Eu = 63, Yb = 70)
1)
Yb2+
2)
Ce2+
3)
Sm2+
4)
Eu2+
Q-2:-
Which of the following structures is similar to graphite?
Q-3:-
An excess of $\text{AgNO}_3$ is added to $100,\text{mL}$ of a $0.01,\text{M}$ solution of dichlorotetraaquachromium(III) chloride, $[\text{Cr}(\text{H}_2\text{O})_4\text{Cl}_2]\text{Cl}$. The number of moles of $\text{AgCl}$ precipitated is:
Q-4:-
A metal has a face-centered cubic (fcc) lattice. The edge length of the unit cell is $404,\text{pm}$. The density of the metal is $2.72,\text{g cm}^{-3}$. The molar mass of the metal is:
(Given: Avogadro’s constant $N_A = 6.02 \times 10^{23},\text{mol}^{-1}$)
Q-5:-
A single compound of the structure

is obtainable from ozonolysis of which of the following cyclic compounds ?
1)

2)

3)

4)

Q-6:-
Propionic acid with Br2 | red P yields a dibromo product. Its structure would be:
1)

2)
CH2Br - CH2 - COBr
3)

4)
CH2 Br - CHBr - COOH
Q-7:-
Of the following sets which one does NOT contain isoelectronic species?
Q-8:-
Q. The equilibrium constants Kp1 and Kp2 for the reactions
X⇌2Y and Z⇌P+Q, respectively, are in the ratio 1:9. If the degree of dissociation of X and Z are equal, the ratio of total pressures at equilibrium is:
Q-9:-
Solubility product of silver bromide is 5.0 × 10-13 . The quantity of potassium bromide (molar mass taken as 120 g mol-1) to be added to 1 litre of 0.05M solution of silver nitrate to start the precipitation of AgBr is
1)
5.0 × 10-8 g
2)
1.2 x 10-10 g
3)
1.2 x 10-9 g
4)
6.2 x 10-5 g
Q-10:-
If an endothermic reaction is non-spontaneous at the freezing point of water and becomes feasible at its boiling point, then
1)
ΔH is −ve,ΔS is +ve
2)
ΔH and ΔS both are +ve
3)
ΔH and ΔS both are - ve
4)
ΔH is +ve,ΔS is −ve
Q-11:-
In an experiment it showed that 10 ml of 0.05M solution of chloride required 10 mL of 0.1 M solution of AgNO3 , which of the following will be the formula of the chloride (X stands for the symbol of the element other than chlorine):
1)
X2Cl
2)
X2Cl2
3)
XCl2
4)
XCl4
Q-12:-
Which of the following acids does not exhibit optical isomerism?
1)
Maleic acid
2)
α - amino acids
3)
Lactic acid
4)
Tartaric acid
Q-13:-
Which of the following cannot form azeotropic mixture?
1)
H2O + C2H5OH
2)
n-Hexane + n-heptane
3)
HNO3 + H2O
4)
None
Q-14:-
Ethanol is manufactured by the fermentation of:
1)
Molasses
2)
Starch
3)
Both a & b
4)
Glucose
Q-15:-
The number of covalent and ionic bonds in the compound $\mathrm{CH_3MgBr}$ are:
Q-16:-
Which among the following contains glycerol and rosin (gum)?
1)
Transparent soaps
2)
Shaving soaps
3)
Medicinal Soaps
4)
Floating soaps
Q-17:-
Which if the following antiseptic can be used as mouthwash or gargles ?
1)
Boric acid
2)
Amyl meta cresol
3)
H2O2
4)
Hg2Cl2
Q-18:-
Coupling reaction involves what mechanism?
1)
nucleophilic substitution
2)
Electrophilic addition
3)
nucleophilic addition
4)
Electrophilic substitution
Q-19:-
X is :
1)
R-H
2) R−NH2+CH4
3) R−NH−CH3
4) R−N=CH2
Q-20:-
Which of the following acid is unstable ?
1)
H2SO4
2)
HNO3
3)
HNO2
4)
HClO3
Q-21:-
Hyperconjugation is also known as :
1)
No bond resonance
2) sp3−p conjugation
3) σ-bond resonance
4)
All of these
Q-22:-
Which of the following reaction proceeds nearly to completion ?
Q-23
The common name of tetrachloromethane is:
1) Chloroform
2) D.D.T
3) Allyl chloride
4) Carbon tetrachloride
Q-24
Copper crystallizes in a face-centered cubic (FCC) lattice with a unit cell length of 361,pm. What is the radius of a copper atom in pm?
1) 108
2) 128
3) 157
4) 181
An element with atomic number 23 belongs to which period?
1)
3rd period
2)
2nd period
3)
5th period
4)
4th period
Q-26:-
As a result of Schottky defect
1)
There is no effect on density
2)
Density increases
3)
Density decreases
4)
All of the above
Q-27:-
The amount of heat evolved when 1 g equivalent of the acid is completely neutralized by a strong base in a dilute solution is called as:
1)
Heat of neutralization of base
2)
Heat of neutralization of acid
3)
Heat of solution
4)
Heat of dilution
Q-28:-
In the reaction
Ph−MgBr+Ph−OH→ Major product formed is:
3)

4)

Q-29:-
Which one of the following methods can be used to obtain highly pure metal which is liquid at room temperature?
1)
Electrolysis
2)
Chromatography
3)
Distillation
4)
Zone refining
Q-30:-
Which of the following particles has more electrons than neutrons
1)
C
2)
F-
3)
O2-
4)
Al3+
Q-31:-
An element of atomic number 29 belongs to
1)
s - block
2)
p - block
3)
d - block
4)
f - block
Q-32:-
Consider the reaction sequence given below
![]()
1)

2)

3)

4)

Q-33:-
Meso compounds are compounds which have chiral centres but also have a mirror image which is super imposable on itself which of the following compound is mesho?
1)

2)

3)

4)

Q-34:-
Which of the following statements is true about the polarity of BeCl2 ?
1)
The molecule is polar due to the difference in electronegativity between Be and Cl.
2)
The molecule is polar due to the lone pair of electrons on the central Be atom.
3)
The molecule is nonpolar due to the symmetric arrangement of the two Be-Cl bonds.
4)
The polarity of the molecule cannot be determined without knowing the bond lengths.
Q-35:-
Which one will have the maximum number of molecules of nitrogen?
1)
14 molecules of nitrogen
2)
1.4 grams of nitrogen
3)
14 grams of nitrogen
4)
14 moles of nitrogen
Q-36:-
A unit cell of a crystal has a body-centered cubic (BCC) lattice with an edge length of 3A. What is the volume of the unit cell in A3 ?
1)
9
2)
27
3)
81
4)
108
Q-37:-
Native iron metal forms a water-soluble complex with a dilute aqueous solution of NaCN in the presence of:
1)
Nitrogen
2)
Oxygen
3)
Carbon dioxide
4)
Argon
Q-38:-
In presence of dil. H2SO4, oxalic acid is oxidized by potassium permanganate. The product that acts as an autocatalyst is
1)
MnSO4
2)
Mn2O3
3)
MnO2
4)
MnO
Q-39:-
The element with highest ionization energy is
1)
He
2)
Ne
3)
Ar
4)
Kr
Q-40:-
In Bosch’s process, Fe2O3 acts as
1)
Catalyst
2)
Promoter
3)
One of the products
4)
One of the reactants
Q-41:-
Which among the following is correct for all the alkali metals?
1)
They readily react with halogens to form ionic halides, M+X−
2)
They react with water to form hydroxide and hydrogen
3)
Their chlorides are deliquescent and crystallise as hydrates
4)
Their nitrates decompose into nitrites and oxygen on heating
Q-42:-
Which of the following statements about the froth flotation process is FALSE?
1)
It is used for the concentration of sulfide ores.
2)
The principle involved is the preferential wetting of ore particles by water.
3)
Air bubbles are introduced to carry the mineral particles to the surface.
4)
Froth stabilisers are used to stabilize the froth.
Q-43:-
Consider the reaction:
$\mathrm{N_2 + 3H_2 \rightarrow 2NH_3}$
$2.5$ moles of $\mathrm{N_2}$ and $4$ moles of $\mathrm{H_2}$ are taken in a $10,\text{L}$ flask at $27^\circ\text{C}$. After complete reaction, $\mathrm{N_2}$ is fully converted into $\mathrm{NH_3}$. Then $5,\text{L}$ of water is added to the flask. The pressure developed in the flask is:
1)
7 atm
2)
7.3 atm
3)
6.95 atm
4)
6 atm
Q-44:-
The first ionization energy of magnesium is lower than the first ionization energy of
1)
Li
2)
Na
3)
Ca
4)
Be
Q-45:-
A first order reaction 80% completed in 80 min. The time taken for reactants to decompose to half of their original amount will be
1)≈50,min
2) ≈60,min
3) ≈40,min
4) ≈34,min
Biology
Q-1:-
FAD + or FADH2
1)
NADH
2)
ATP
3)
H2O
4)
ATP
Q-2:-
In a testcross involving F1 dihybrid flies, more parental-type offspring were produced than the recombinant-type offspring. This indicates:
1) The two genes are located on two different chromosomes.
2) Chromosomes failed to separate during meiosis.
3) The two genes are linked and present on the same chromosome.
4) Both of the characters are controlled by more than one gene.
Q-3:-
Which one of the following statements is not true?
Q-4:-
Lack of relaxation between successive stimuli in sustained muscle contraction is known as:
Q-5:- Which of the following is wrong for viruses
All are parasites
1)
All of them have helical symmetry
2)
They have the ability ot synthesize nucleic acids and proteins
3)
Antibiotics have no effect on them
4)
All of them have helical symmetry
Q-6:-
Which of the following pairs of organelles does not contain DNA?
Q-7:-
Flat worms
1)
Sponges
2)
Ctenophores
3)
Corals
4)
Flat worms
Q-8:-
Following statements describe the characteristics of the enzyme Restriction Endonuclease. Identify the incorrect statement.
Q-9:-
The centriole undergoes duplication during:
Q-10:-
Which stage of meiotic prophase shows terminalization of chiasmata as its distinctive feature?
Q-11:-
What are the two basic categories of ecosystems?
Inland and outland ecosystem
1)
Complex and non-complex ecosystem
2)
Estuary and forest ecosystem
3)
Terrestrial and aquatic ecosystem
4)
Terrestrial and aquatic ecosystem
Q-12:- Ecosystem productivity is related to:
productivity
1)
Energy flow
2)
Nutrient cycling
3)
All of these
4)
All of these
Q-13:- Sharks belong to which group?
1)
Lizard
2)
urodela
3)
Urochordata
4)
Lizard
Q-14:-
Which of the following is a vector-borne disease?
1)
Amoebiasis
2)
Typhoid
3)
Pneumonia
4)
Filariasis
Q-15:-
Metabolic processes
1)
Provide energy
2)
Prevents dehydration
3)
More than one correct answer
4)
More than one correct answer
Q-16:-
Cardiac region is a part of which organ?
1)
fundus
2)
pylorus
3)
Body
4)
fundus
Q.17: The human skeleton consists of:
Q-18:-
Blood plasma mainly consists of:
Q-19:- Urine
is stored in the urinary bladder
1)
is reabsorbed into the blood
2)
gets collected in the renal pelvis
3)
is lost as sweat
4)
is reabsorbed into the blood
Q-20 Insulin is secreted by which of the following cells?
Q. 21 Which condition is caused by reduced blood supply to the heart muscles?
Q 22 Gastric juice is ______ in nature, while pancreatic juice is ______ in nature.
Q-23:-
Which of the following RNAs is not required for the synthesis of protein?
Q-24:-
The clitellum in an earthworm is present in which segments?
Q-25:-
Phloem parenchyma is also known as:
Q-26:-
The production of gametes by the parents, the formation of zygotes, and the F1 and F2 plants can be understood from a diagram called:
Q-27:-
Assertion (A): A person goes to a high altitude and experiences 'altitude sickness' with symptoms like breathing difficulty and heart palpitations.
Reason (R): Due to low atmospheric pressure at high altitudes, the body does not get sufficient oxygen.
In light of the above statements, choose the correct answer from the options given below.
Q-28:-
Which of the following organisms do not produce oxygen?
Q-29:-
Which sequence correctly represents the changes during ageing of stored blood?
Q-30:-
Interferons act by:
Q-31:- Which of the following statements about inbreeding are correct?
Inbreeding increases homozygosity
1)
Inbreeding is essential to evolve pure lines in any animal
2)
Inbreeding selects harmful recessive genes that reduce fertility and productivity.
3)
Inbreeding helps in the accumulation of superior genes and the elimination of undesirable genes.
4)
Inbreeding helps in the accumulation of superior genes and the elimination of undesirable genes.
Q-32:- Which of the following features are found in cartilaginous fishes?
Placoid scale
1)
Ventral mouth
2)
Ctenoid scale and ventral mouth
3)
Placoid scale and ventral mouth
4)
Placoid scale and ventral mouth
Q-33:- Reserve food materials in plants and animals are respectively:
Starch; Glucose
1)
Floridian Starch; Amylopectin and Glycogen
2)
Glucose; Amylopectin and Glycogen
3)
Amylopectin; Monosaccharides
4)
Floridian Starch; Amylopectin and Glycogen
Q-34:-
Cladode is a type of:
1)
Phylloclade
2)
Phyllode
3)
Bulb
4)
Phylloclade
Q-35:- Female genital pores in an earthworm are present on which segments?
10th and 11th segment
1)
12th and 13th segment
2)
14th and 16th segment
3)
18th and 19th segment
4)
Q-36:-
High water potential in phloem at the source leads to:
Q-37:-
Substances that are oxidized during respiration are called:
Q-38:-
In which phase of the growth curve does rapid growth occur?
Q-39:-
Submandibular lymph nodes are located:
Q-40:-
Pancreatic duct and bile duct open into the duodenum through the:
Q-41:- G-cells of the stomach are responsible for secreting:
Q-42:-
Which of the following digestive secretions are released into the small intestine?
Q-43:-
Formed elements of blood include:
Q-44:-
Entry of more than one sperm into an ovum is called:
Q-45:-
Which organism causes syphilis?
Also Check:
The NEET 2026 Mock Test Day 1 answer keys are now available on the same day, allowing you to analyse your performance immediately after attempting the test.
| Q.NO. (Physics) | Answer KEY | Q.NO (Chemistry) | Answer Key | Q.NO. (Biology) | Answer Key | Q. NO. | Answer Key |
| 1 | 4 | 1 | 1 | 1 | 3 | 46 | 1 |
| 2 | 4 | 2 | 2 | 2 | 3 | 47 | 1 |
| 3 | 4 | 3 | 2 | 3 | 1 | 48 | 1 |
| 4 | 3 | 4 | 4 | 4 | 3 | 49 | 4 |
| 5 | 3 | 5 | 3 | 5 | 2 | 50 | 3 |
| 6 | 2 | 6 | 3 | 6 | 3 | 51 | 1 |
| 7 | 1 | 7 | 3 | 7 | 1 | 52 | 3 |
| 8 | 3 | 8 | 2 | 8 | 2 | 53 | 4 |
| 9 | 4 | 9 | 3 | 9 | 1 | 54 | 4 |
| 10 | 1 | 10 | 2 | 10 | 4 | 55 | 3 |
| 11 | 1 | 11 | 3 | 11 | 4 | 56 | 1 |
| 12 | 1 | 12 | 1 | 12 | 4 | 57 | 2 |
| 13 | 1 | 13 | 2 | 13 | 2 | 58 | 2 |
| 14 | 3 | 14 | 4 | 14 | 1 | 59 | 4 |
| 15 | 3 | 15 | 1 | 15 | 4 | 60 | 1 |
| 16 | 1 | 16 | 2 | 16 | 2 | 61 | 4 |
| 17 | 1 | 17 | 2 | 17 | 4 | 62 | 3 |
| 18 | 2 | 18 | 4 | 18 | 4 | 63 | 1 |
| 19 | 3 | 19 | 3 | 19 | 2 | 64 | 2 |
| 20 | 3 | 20 | 3 | 20 | 2 | 65 | 2 |
| 21 | 3 | 21 | 4 | 21 | 1 | 66 | 1 |
| 22 | 1 | 22 | Not Found | 22 | 2 | 67 | 4 |
| 23 | 4 | 23 | 4 | 23 | 1 | 68 | 2 |
| 24 | 3 | 24 | 2 | 24 | 4 | 69 | 1 |
| 25 | 4 | 25 | 4 | 25 | 2 | 70 | 3 |
| 26 | 1 | 26 | 3 | 26 | 3 | 71 | 3 |
| 27 | 2 | 27 | 2 | 27 | 1 | 72 | 4 |
| 28 | 3 | 28 | 2 | 28 | 2 | 73 | 3 |
| 29 | 1 | 29 | 3 | 29 | Not Found | 74 | 3 |
| 30 | 3 | 30 | 3 | 30 | 4 | 75 | 1 |
| 31 | 2 | 31 | 3 | 31 | 4 | 76 | 1 |
| 32 | 2 | 32 | 1 | 32 | 4 | 77 | 1 |
| 33 | 1 | 33 | 4 | 33 | 2 | 78 | 1 |
| 34 | 2 | 34 | 3 | 34 | 2 | 79 | 1 |
| 35 | 4 | 35 | 4 | 35 | 2 | 80 | 3 |
| 36 | 3 | 36 | 4 | 36 | 2 | 81 | 4 |
| 37 | 3 | 37 | 2 | 37 | 4 | 82 | 2 |
| 38 | 2 | 38 | 1 | 38 | 2 | 83 | 1 |
| 39 | 2 | 39 | 1 | 39 | 4 | 84 | 2 |
| 40 | 1 | 40 | 1 | 40 | 2 | 85 | 3 |
| 41 | 3 | 41 | 2 | 41 | 3 | 86 | 2 |
| 42 | 1 | 42 | 2 | 42 | 4 | 87 | 4 |
| 43 | 3 | 43 | 2 | 43 | 2 | 88 | 2 |
| 44 | 4 | 44 | 4 | 44 | 3 | 89 | 1 |
| 45 | 2 | 45 | 4 | 45 | 1 | 90 | 4 |
After checking your answers, follow a focused analysis approach:
This same-day analysis strategy helps convert mistakes into learning and significantly improves your NEET score.
In the final days before NEET, your score depends more on how well you perform under exam conditions than how much you study. This mock test is designed to sharpen your execution and maximise your final score.
Consistent practice with this mock creates a performance improvement loop, helping you turn preparation into marks.
In the final stretch before NEET, following a structured daily routine can make a significant difference in your score. The goal is to combine practice, analysis, and revision within the same day for maximum retention.
This routine creates a same-day improvement cycle, helping you quickly fix mistakes, strengthen concepts, and improve your NEET score efficiently.
Avoiding mock tests in the final phase of NEET preparation can seriously impact your performance. Without regular practice under exam conditions, even strong concepts may not translate into good scores.
Skipping mock tests can lead to:
NEET is not just a test of knowledge - it’s a test of performance, speed, and accuracy. Regular mock practice ensures you are fully prepared to handle the real exam confidently.
Also Read:
Frequently Asked Questions (FAQs)
After 10 AM and before 5 PM to simulate real NEET exam conditions effectively.
The answer key will be available after 5 PM on the same day.
Yes, it follows the latest NCERT-based NEET 2026 exam pattern.
Yes, consistent mock practice significantly improves accuracy, speed, and exam confidence.
Ideally 7–10 full-length mocks in the final preparation phase
On Question asked by student community
Hello Dear Student,
No, NEET is not compulsory for Bachelor of Physiotherapy (BPT) admissions for the 2026–27 academic session.
You can check, find and access more information here:
https://news.careers360.com/physiotherapy-neet-admission-1yr-internship-bpt-dr-title-allied-health-sciences-paramedical-courses-revamp-syllabus-ncahp-mohfw
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-mds-2025-seat-allotment-result
Hope it helps!
Hello Dear Student,
With a NEET MDS 2026 score of 503 and an AIR of around 4600, getting a government seat in highly competitive branches such as Orthodontics, Conservative Dentistry & Endodontics, or Prosthodontics may be difficult, as these specialties usually close at much better ranks.
However, you still have
Hello Dear Student,
Haryana NEET MDS 2026 counselling registrations are expected to begin in July 2026. The official schedule will be released by the Department of Medical Education and Research (DMER) Haryana after the national MCC MDS counselling process commences.
You can check, find and access more information here:
https://medicine.careers360.com/articles/haryana-neet-mds-counselling
Hi!
Given below is the link to access the NEET 2026 Physics Question Paper:
https://medicine.careers360.com/articles/neet-physics-question-paper
You can check the link below, apply relevant filters, find suitable resources related to NEET that will boost your overall preparation:
https://www.careers360.com/download/neet-sample-papers-previous-years
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs