JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
NEET 2026 Mock Test Day 3 is LIVE now (10 AM–5 PM)! This is the stage where your score starts improving—not just your preparation. Attempt today’s full-length paper under real exam conditions and analyse your performance with the same-day answer key and detailed solutions. If your marks fluctuated in Day 1 and Day 2, this is the test where your performance starts stabilising.
This Story also Contains

At this stage, improvement comes from performance—not just preparation. Day 3 of this NEET mock test series is designed to strengthen your accuracy, speed, and time management through NCERT-based, exam-level questions from Physics, Chemistry, and Biology. Practising full-length tests daily and analysing them the same day is one of the most effective ways to maximise your NEET score in the last stretch before the NEET exam.
This full-length Day 3 mock test is designed to simulate the real exam environment and help you evaluate your performance under actual NEET conditions. At this stage, consistent full-length practice is what separates improving scores from stagnant ones. This method is widely followed by top-performing NEET aspirants to build accuracy and confidence under pressure.
Full-length NEET mock test (180 questions)
Covers Physics, Chemistry, and Biology
Based on NCERT concepts and previous year trends
Aligned with the real NEET difficulty level
Includes same-day answer key and detailed solutions
If your score did not improve in Day 1 or Day 2, Day 3 is where you start fixing mistakes and stabilising accuracy. Focus on analysis today—not just attempts. This NEET 2026 daily mock test series is exactly what toppers follow in the final days.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
To maximise your score improvement with NEET 2026 Mock Test Day 3, follow a strict exam-oriented approach instead of casual solving. Your focus should be on performance, not just completion.
Attempt the full paper before 10 AM to match actual exam timing
Complete the test within 3 hours without interruption
Avoid checking answers or pausing during the attempt
Review the answer key after 5 PM only
Analyse mistakes deeply and revise weak topics on the same day
Physics → 50 minutes
Chemistry → 45 minutes
Biology → 85 minutes
This same-day practice and analysis cycle is one of the most effective strategies used by NEET toppers to improve accuracy, reduce errors, and stabilise scores in the final days before the exam.
This is not just practice—it is your real NEET 2026 exam simulation. Treat this attempt seriously, because how you perform today reflects your exam readiness.
Also Attempt:
Day 1: Understanding the NEET Exam Pattern and baseline performance
Day 2: Improving accuracy and adapting strategy
Day 3: Stabilising your score by correcting repeated mistakes
This is the transition phase where preparation starts converting into marks. Your score in this test is a strong indicator of your actual NEET 2026 performance.
How you perform in this test closely reflects your expected NEET 2026 score range.
Instructions:
Attempt all questions in a strictly timed environment (3 hours) to simulate the real NEET exam
Do not pause the test or refer to notes during the attempt
Avoid checking answers while solving the paper
Complete the full mock before reviewing the answer key
Focus on accuracy, time management, and consistency under pressure
Treat this as your real exam attempt—your performance today directly reflects your NEET 2026 readiness.
The codon 'AUG' codes for methionine and phenylalanine.
Statement II :
'AAA' and 'AAG are both codons that code for the amino acid lysine.
In light of the above statements, choose the correct answer from the options given below.
(1) Both Statement I and Statement II are true
(2) Both Statement I and Statement II are false
(3) Statement I is correct but Statement II is false
(4) Statement I is incorrect but Statement II is true.
| Column I | Column II |
|---|---|
| 1. Mons pubis | A. Fleshy folds of tissue |
| 2. Labia majora | B. Paired folds of tissue |
| 3. Labia minora | C. Finger-like structures |
| 4. Clitoris | D. A cushion of fatty tissue |
Q-1: Two cars moving in opposite directions approach each other with speeds of 22 m/s and 16.5 m/s respectively. The driver of the first car blows a horn having a frequency of 400 Hz. The frequency heard by the driver of the second car is [velocity of sound 340 m/s]:
Q-2: In Young’s double slit experiment, the distance between slits and the screen is 1.0 m and monochromatic light of 600 nm is used. A person near the slits observes the fringe pattern. When slit separation is varied, the pattern disappears for a distance d0. If angular resolution of the eye is (1°/60), find d0:
Q-3: A pendulum clock loses 12 s/day at 40°C and gains 4 s/day at 20°C. Find the temperature at which it shows correct time and coefficient of linear expansion (α):
Q-4: Two radioactive nuclei P and Q decay into stable nucleus R. Initially, P = 4N and Q = N. Half-life of P is 1 min and Q is 2 min. When P and Q become equal, number of R nuclei is:
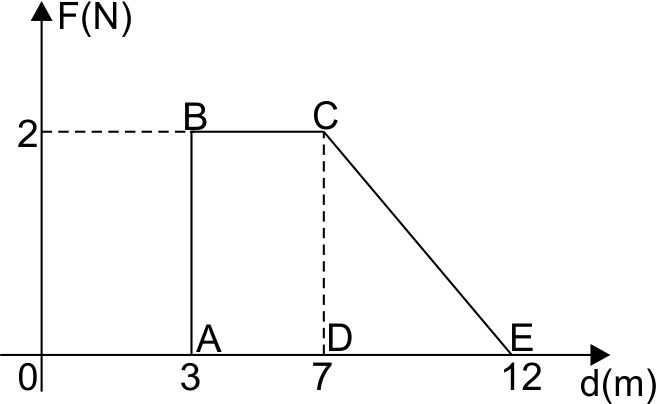
Q-5: Force F varies with distance as shown. Work done during 12 m displacement is:
Q-6: For transistor action:
Correct pair:
Q-7: A red LED emits 0.1 W uniformly. Electric field amplitude at 1 m is:
Q-8: A diatomic gas has rms speed 1930 m/s at room temperature. Identify the gas:
Q-9: Dielectric constant varies as K(x) = K₀ + λx. Capacitance relation with vacuum capacitance C₀ is:
Q-10: Two wires (same material) in parallel. Length ratio = 4/3, radius ratio = 2/3. Current ratio:
Q-11: Two coaxial solenoids wound over a pipe (area = 10 cm2, length = 20 cm) have 300 and 400 turns. Mutual inductance is (μ0 = 4π × 10-7 TmA-1):
Q-12: A mass m moving along x-axis with velocity V collides and sticks to mass 3m moving upward (y-axis) with velocity 2V. Final velocity is:
Q-13: A rope is wound around a hollow cylinder (mass 3 kg, radius 0.4 m). Force applied = 30 N. Angular acceleration:
Q-14: Threshold frequency = 3.3 × 1014 Hz. Incident frequency = 8.2 × 1014 Hz. Cut-off voltage:
Q-15: A solenoid (2×104 turns/m) carries current reducing from 4 A to 0 in 0.05 s. A 100-turn coil (radius 0.01 m) is placed inside. Resistance = 10π2 Ω. Total charge:
Q-16: EM wave enters a medium. Electric fields are given by E1 = E0 sin(ωt - kx) and E2 = E0 sin(ωt - kx/2). Ratio of relative permittivities (εr1/εr2):
Q-17: If de-Broglie wavelengths of proton and α-particle are equal, ratio of velocities (vp : vα):
Q-18: A particle moves north at 50 m/s and after 10 s moves west at same speed. Motion is:
Q-19: Electron and proton move equal distance in same electric field. Ratio of times t1/t2:
Q-20: Angle of dip gives:
Q-21:
A block of mass $m$ is moving with velocity $v$ on a rough horizontal surface and comes to rest after traveling a distance $d$. The coefficient of friction between the block and surface is:
Q-22: Angular momentum of satellite (mass m, orbit radius R0):
Q-23: Two cars with accelerations a1 and a2 start from rest. Velocity difference at finish:
Q-24: Two waves given by y1 = a sin(ωt) and y2 = b sin(ωt + φ). Intensity ratio Imax/Imin is:
Q-25:
The de Broglie wavelength of a particle is $\lambda$. If its kinetic energy is doubled, the new wavelength becomes:
Q-26: Branch studying motion without forces:
Q-27: Correct statement:
Q-28: Work against gravity vs potential energy:
Q-29: Electron emission due to strong electric field:
Q-30: Nucleus has 30 neutrons. Radius ratio with He nucleus = 141/3. Atomic number:
Q-31: A charge $q$ is placed at the centre of a cube. What is the electric flux through one face of the cube?
Q-32: Velocity v = αt2. Ratio (a/v) at t = 2 s:
Q-33: x(t) = bt + at2. Velocity when acceleration = 0:
Q-34: Charge = -2 μC, protons = 2.5×1013. Number of electrons:
Q-35: Three capacitors (1 μF) in parallel + one in series:
Q-36: Escape velocity from midpoint Earth-Moon (Distance d):
Q-37: Rate of area swept in elliptical orbit is proportional to:
Q-38: Time difference for beams entering water (refractive index μ):
Q-39: Relation between work done and degree of freedom (n):
Q-40: Isothermal expansion, pressure change ratio:
Q-41: For diatomic gas Q:ΔU:W ratio:
Q-42: Wave equation phase at t=7.5 s:
Q-43: Electron-hole drift velocity ratio (ve/vh):
Q-44: Wire vibration frequency when tension is increased by 44%:
Q-45: In a certain region of space with a volume of 0.2 m3, the electric potential is found to be 5V throughout. The magnitude of the electric field in this region is:
"Q-1:- Pick out the correct statement with respect to $\left[\mathrm{Mn}(\mathrm{CN})_6\right]^{3-}$ :
1) It is $\mathrm{sp}^3 \mathrm{~d}^2$ hybridised and octahedral
2) It is $\mathrm{sp}^3 \mathrm{~d}^2$ hybridised and tetrahedral
3) It is $\mathrm{d}^2 \mathrm{sp}^3$ hybridised and octahedral
4) It is $\mathrm{dsp}^2$ hybridised and square planar
" "Q-2:- Which of the following complexes is used to be as anticancer agent ?
1) mer - [Co (NH3)3 Cl3]
2) cis - [Pt Cl2 (NH3)2]
3) cis - K2[Pt Cl2 Br2]
4) Na2CoCl4
" "Q-3:- Which of the statements given below is incorrect?
1) $\mathrm{Cl}_2 \mathrm{O}_7$ is an anhydride of perchloric acid.
2) $\mathrm{O}_3$ molecule is bent
3) ONF is isoelectronic with O2N−
4) OF2 is an oxide of fluorine
" "Q-4:- Which one of the following statements is not true regarding (+) Lactose?
1) (+) Lactose, C12H22O11 contains 8 - OH groups
2) On hydrolysis (+) Lactose gives equal amount of D (+) glucose and D(+) galactose.
3) (+) Lactose is a -glycoside formed by the union of molecule of D(+) glucose and a molecule of D (+) galactose
4) (+) Lactose is a reducing sugar and does not exhibit mutarotation
" "Q-5:- The correct order of the decreasing ionic radii among the following isoelectronic species is :
1) K+ > Ca2+ > Cl- > S2-
2) Ca2+ > K+ > S2- > Cl-
3) Cl- > S2- > Ca2+ > K+
4) S2- > Cl- > K+ > Ca2+
Q-6:- For the compounds CH3Cl, CH3Br, CH3I, and CH3F,
the correct order of increasing C-halogen bond length is :
1) CH3F < CH3Cl < CH3Br < CH3I
2) CH3F < CH3Br < CH3Cl < CH3I
3) CH3F < CH3I < CH3Br < CH3Cl
4) CH3Cl < CH3Br < CH3F < CH3I
Q-7:- The gas leaked from a storage tank of the Union Carbide plant in Bhopal gas tragedy was:
1) Phosgene
2) Methyl isocyanate
3) Methylamine
4) Ammonia
Q-8:- When rain is accompanied by a thunderstorm, the collected rain water will have a value
1) slightly lower than that of rain water without thunderstorm
2) lightly higher than that when the thunderstorm is not there
3) uninfluenced by occurrence of thunderstorm
4) which depends on the amount of dust in air.
Q-9:- Bakelite is obtained from phenol by reaction with
1) HCHO
2) $\left(\mathrm{CH}_2 \mathrm{OH}\right)_2$
3) $\mathrm{CH}_3 \mathrm{CHO}$
4) $\mathrm{CH}_3 \mathrm{COCH}_3$
Q-10:- Acetyl bromide reacts with excess of $\mathrm{CH}_3 \mathrm{MgI} {\text {followed by treatment with }}$ a saturated solution of $\mathrm{NH}_4 \mathrm{Cl}$ gives
1) acetone
2) acetamide
3) 2-methyl-2-propanol
4) acetyl iodide.
Q-11:- In which of the following arrangements the given sequence is not strictly according to the property indicated against it?
Q-12:- Which of the following have Schottky defect ?
1) ZnS
2) NaCl
3) CuBr
4) AgCl
Q-13:- Which of the following compounds can form a zwitterion?
1) Benzoic acid
2)
Acetanilide
3)
Aniline
4)
Glycine
" "Q-14:-
Propan-2-ol upon treatment with which reagent will give acetone?
1)
$\mathrm{KMnO}_4 / \mathrm{H}_2 \mathrm{SO}_4$
2)
PDC
3)
$\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7 / \mathrm{H}_2 \mathrm{SO}_4$
4)
$\mathrm{KMnO}_4 / \mathrm{NH}_4 \mathrm{Cl}$
" "Q-15:-
Which of the following amines can be obtained by the reduction of oximes using Na in ethanol solvent?
Q-16:-
The most negative electron gain enthalpy of group 16 element is
1)
S
2)
Se
3)
Te
4)
Po
" "Q-17:- Which of the following sets of molecules will have zero dipole moment?
1) Boron trifluriode, beryllium difluoride, carbon dioxide, 1,4- dichlorobenzene
2) Ammonia, breyllium difluriode ,water, 1,4- dichlorobenzene
3) Boron trifluoride, hydrogen fluoride, carbon dioxide, 1,3- dichlorobenzene
4) Nitrogen trifluoride
Q-18:- A tertiary butyl carbocation is more stable than a secondary butyl carbocation because of which of the following?
1) Hyperconjugation
2) -I effect of -CH3 groups
3) +R effect of $\mathrm{CH}_3$ groups
4) -R effect of $-\mathrm{CH}_3$ groups
" "Q-19:- Which of the following alkanes cannot be made in good yield by the Wurtz reaction?
1) n- butane
2) n- Hexane
3) 2,3- Dimethylbutane
4) n- Hepatne
Q-20:-
The compound used in the treatment of lead poisoning is:
1)
D-penicillamine
2)
Desferrioxamine B
3)
Cis-platin
4)
EDTA
" "Q-21:-
Which hydride is neutral?
1)
H2S
2)
H2O
3)
H2Se
4)
H2Te
Q-22:-
The IUPAC name of an element with atomic number 104 is:
1)
Unnilunium
2)
Unniltrium
3)
Unnilquadium
4)
Unnilbium
Q-23:-
Octet rule cannot be applied to the non- metals after
1)
Carbon
2)
Silicon
3)
Oxygen
4)
Nitrogen
Q-24:-
Which of the following is an amorphous solid?
1)
Glass
2)
NaCl
3)
AgCl
4)
ZnS
Q-25:-
Which one of the following statements regarding a dynamic equilibrium is false?
1)
At equilibrium, there is no net change in the system
2)
At equilibrium, the concentration of reactants and products stays the same.
3)
At equilibrium, the forward and back reactions cease to occur
4)
At equilibrium, the rates of the forward and back reactions are identical
Q-26:-
A metal (A) on heating in nitrogen gas gives compound B. B on treatment with gives a colourless gas which when passed through solution gives a dark blue-violet coloured solution. A and B respectively, are :
Q-27:-
Consider the following plots of rate constant versus $\frac{1}{\mathrm{~T}}$ for four different reactions. Which of the following orders is correct for the activation energies of these reactions ?
1)
$E_b>E_a>E_d>E_c$
2)
$E_a>E_c>E_d>E_b$
3)
$E_b>E_d>E_c>E_a$
4)
$E_c>E_a>E_d>E_b$
" "Q-28:-
Match the following aspects with the respective metal.
| Aspects | Metal |
| (a) The metal which reveals a maximum number of oxidation states | (i) Scandium |
| (b) The metal although placed in 3d block is considered not as transition element | (ii) Copper |
| (c) The metal which does not exhibit variable oxidation states | (iii) Manganese |
| (d) The metal which in +1 oxidation state in aqueous solution undergoes disproportionation | (iv) Zinc |
Select the correct option :
1)
(a) - (iii) (b) - (iv) (c) - (i) (d) -(ii)
2)
(a) - (iii) (b) - (i) (c) - (iv) (d) -(ii)
3)
(a) - (ii) (b) - (iv) (c) - (i) (d) -(iii)
4)
(a) - (i) (b) - (iv) (c) - (ii) (d) -(iii)
Q-29:-
Statement A: Helium belongs to s block but it is placed in p block .
Statement B: Helium has completely filled valence shell.
1)
Both statements are correct and statement B is correct explanation for statement A.
2)
Both statements are correct and statement B is not a correct explanation for statement A.
3)
Statement A is correct and statement B is incorrect.
4)
Statement A is incorrect and statement B is correct.
Q-30:-
Elements X and Y combine to form two compounds XY and $X Y_2$ . Find the atomic weight of X and Y, if the weight of 0.1 moles of XY is 15g and 0.1 moles of $X Y_2$ is 25g.
1)
X = 50g , Y = 100g
2)
X = 100g , X = 50g
3)
X = 25g , Y = 50g
4)
X = 50g , Y = 25g
Q-31:-
Assertion: Carbon dioxide $\mathrm{CO}_2$ is a greenhouse gas.
Reason: Carbon dioxide can absorb and re-emit infrared radiation, which can trap heat in the atmosphere.
1)
Both assertion and reason are true, and the reason is the correct explanation of the assertion.
2)
Both assertion and reason are true, but the reason is not the correct explanation of the assertion.
3)
The assertion is true, but the reason is false.
4)
The assertion is false, but the reason is true.
Q-32:-
Which of the following gaseous air pollutants is not emitted directly by human activities, but is formed in the atmosphere as a result of chemical reactions?
1)
Sulfur dioxide $\left(\mathrm{SO}_2\right)$
2)
Carbon monoxide $(\mathrm{CO})$
3)
Nitrogen oxide $\left(N O_x\right)$
4)
Ozone $\left(O_3\right)$
" "Q-33:-
Assertion: A solution with a higher molality will always exhibit a larger boiling point elevation than a solution with a lower molality.
Reasoning: The boiling point elevation of a solution is directly proportional to the molality of the solution.
1)
Both assertion and reasoning are true, and the reasoning is the correct explanation of the assertion.
2)
Both assertion and reasoning are true, but the reasoning is not the correct explanation of the assertion.
3)
Assertion is true, but reasoning is false.
4)
Assertion is false, but reasoning is true.
Q-34:-
What is the Zeta potential of a colloidal particle?
1)
The potential difference across the Electric Double Layer.
2)
The charge on the surface of the colloidal particle.
3)
The size of the colloidal particle.
4)
The concentration of the colloidal particle in the solution
Q-35:-
Which of the following statements is true regarding adsorption isotherms?
1)
They show the variation in the amount of adsorbent with pressure at constant temperature
2)
They show the variation in the amount of adsorbent with temperature at constant pressure
3)
They show the variation in the amount of adsorbate with pressure at constant temperature
4)
They show the variation in the amount of adsorbate with temperature at constant pressure
" "Q-36:-
Which of the following statements is true regarding the constants k and n in the Freundlich adsorption isotherm?
1)
They are independent of the nature of the adsorbent and the gas.
2)
They are independent of the temperature.
3)
They depend on the nature of the adsorbent and the gas.
4)
They depend on the temperature.
" "Q-37:-
Assertion: Alumina is obtained from bauxite through the Bayer process.
Reasoning: The Bayer process involves the leaching of bauxite with sodium hydroxide solution, followed by the precipitation of aluminum hydroxide, which is then calcined to obtain alumina.
1) Both the assertion and reasoning are true, and the reasoning is the correct explanation of the assertion.
2) Both the assertion and reasoning are true, but the reasoning is not the correct explanation of the assertion.
3) The assertion is true, but the reasoning is false.
4) Both the assertion and reasoning are false.
Q-38:-
On boiling hard water calcium bicarbonate precipitates out as calcium carbonate but magnesium bicarbonate precipitate out as magnesium hydroxide because
1) The solubility of $\mathrm{CaCO}_3$ is more than $\mathrm{MgCO}_3$
2) The solubility of $\mathrm{MgCO}_3$ is more than that of $\mathrm{CaCO}_3$
3) The solubility of $\mathrm{Mg}(\mathrm{OH})_2$ is less than $\mathrm{Ca}(\mathrm{OH})_2$
4) Both b and c
Q-39:-
At and 760 torr, 40 % of molecules are dissociated in a given sample of gas. What is the density of the given mixture at equilibrium?
1) 1.48 g/L
2) 2.66 g/L
3) 3.12 g/L
4) 2.25 g/L
Q-40:-
Which option correctly describes the relationship between $k_1, k_2, k_3$ and $k_4$, where the rate expression for the reaction
$\mathrm{NO(g) + H_2(g) \rightarrow \tfrac{1}{2}N_2(g) + H_2O(g)}$ is expressed as:
$\dfrac{d[N_2]}{dt} = k_1[NO][H_2], \quad \dfrac{d[H_2O]}{dt} = k_2[NO][H_2]$
$-\dfrac{d[NO]}{dt} = k_3[NO][H_2], \quad -\dfrac{d[H_2]}{dt} = k_4[NO][H_2]$
Q-41:-
Which of the following alkaline earth metal hydroxides is amphoteric in nature-
1) $\mathrm{Be}(\mathrm{OH})_2$
2) $\mathrm{Ca}(\mathrm{OH})_2$
3) $\mathrm{Sr}(\mathrm{OH})_2$
4) $\mathrm{Ba}(\mathrm{OH})_2$
Q-42:- For an ideal gas, which of the following statements is correct?
Q-43:-
The thermal decomposition of a compound follows second-order kinetics. If the rate of reaction is $R$ when the concentration of the reactant is $[A]$, what will be the rate of reaction when the concentration is increased to three times its initial value?
Q. 44 The number of unpaired electrons in $\mathrm{[Fe(CN)_6]^{4-}}$ is:
Q. 45 Which of the following reagents converts a primary alcohol into an aldehyde without over-oxidation?
The NEET 2026 Mock Test Day 3 answer key and detailed solutions are now available and updated on the same day. Use this to evaluate not just your score, but how well your performance is stabilising after the first two mock tests.
|
Q.NO. (Biology) |
Answer Key |
Q. NO. |
Answer Key |
Q.NO. (Physics) |
Answer KEY |
Q.NO (Chemistry) |
Answer Key |
|
1 |
4 |
46 |
4 |
1 |
4 |
1 |
3 |
|
2 |
1 |
47 |
1 |
2 |
2 |
2 |
2 |
|
3 |
1 |
48 |
3 |
3 |
1 |
3 |
4 |
|
4 |
4 |
49 |
2 |
4 |
4 |
4 |
4 |
|
5 |
4 |
50 |
2 |
5 |
1 |
5 |
4 |
|
6 |
2 |
51 |
4 |
6 |
3 |
6 |
1 |
|
7 |
3 |
52 |
4 |
7 |
2 |
7 |
2 |
|
8 |
1 |
53 |
4 |
8 |
1 |
8 |
1 |
|
9 |
2 |
54 |
4 |
9 |
3 |
9 |
1 |
|
10 |
Not Found |
55 |
4 |
10 |
2 |
10 |
3 |
|
11 |
4 |
56 |
4 |
11 |
1 |
11 |
2 |
|
12 |
3 |
57 |
3 |
12 |
1 |
12 |
2 |
|
13 |
3 |
58 |
1 |
13 |
2 |
13 |
4 |
|
14 |
1 |
59 |
1 |
14 |
1 |
14 |
2 |
|
15 |
1 |
60 |
3 |
15 |
2 |
15 |
1 |
|
16 |
2 |
61 |
3 |
16 |
4 |
16 |
1 |
|
17 |
4 |
62 |
1 |
17 |
1 |
17 |
1 |
|
18 |
3 |
63 |
3 |
18 |
1 |
18 |
1 |
|
19 |
3 |
64 |
3 |
19 |
2 |
19 |
3 |
|
20 |
2 |
65 |
3 |
20 |
4 |
20 |
4 |
|
21 |
2 |
66 |
3 |
21 |
1 |
21 |
2 |
|
22 |
1 |
67 |
1 |
22 |
1 |
22 |
3 |
|
23 |
4 |
68 |
2 |
23 |
1 |
23 |
2 |
|
24 |
4 |
69 |
3 |
24 |
2 |
24 |
1 |
|
25 |
4 |
70 |
2 |
25 |
2 |
25 |
3 |
|
26 |
4 |
71 |
2 |
26 |
3 |
26 |
2 |
|
27 |
4 |
72 |
3 |
27 |
1 |
27 |
4 |
|
28 |
1 |
73 |
1 |
28 |
1 |
28 |
1 |
|
29 |
3 |
74 |
1 |
29 |
3 |
29 |
1 |
|
30 |
3 |
75 |
3 |
30 |
2 |
30 |
1 |
|
31 |
4 |
76 |
1 |
31 |
2 |
31 |
1 |
|
32 |
1 |
77 |
3 |
32 |
4 |
32 |
4 |
|
33 |
4 |
78 |
2 |
33 |
3 |
33 |
1 |
|
34 |
1 |
79 |
3 |
34 |
3 |
34 |
1 |
|
35 |
1 |
80 |
4 |
35 |
4 |
35 |
3 |
|
36 |
4 |
81 |
1 |
36 |
4 |
36 |
3 |
|
37 |
2 |
82 |
4 |
37 |
4 |
37 |
1 |
|
38 |
4 |
83 |
4 |
38 |
4 |
38 |
4 |
|
39 |
2 |
84 |
2 |
39 |
4 |
39 |
2 |
|
40 |
2 |
85 |
1 |
40 |
3 |
40 |
2 |
|
41 |
3 |
86 |
3 |
41 |
3 |
41 |
1 |
|
42 |
4 |
87 |
2 |
42 |
3 |
42 |
2 |
|
43 |
3 |
88 |
4 |
43 |
3 |
43 |
3 |
|
44 |
4 |
89 |
3 |
44 |
4 |
44 |
1 |
|
45 |
3 |
90 |
1 |
45 |
4 |
45 |
3 |
At this stage, your focus should be on identifying whether your mistakes are reducing and your accuracy is becoming consistent.
After checking the answer key, focus on:
Tracking improvement vs Day 1 and Day 2 performance
Identifying repeated mistakes that still persist
Analysing accuracy across Physics, Chemistry, and Biology
Checking if errors are due to concept gaps or exam pressure
Revising weak areas immediately for same-day correction
In the last few days before NEET 2026, score improvement comes from execution—not just revision. This Day 3 test is designed to train your brain for real exam performance by combining accuracy, speed, and pressure handling in a single attempt.
This full-length NEET mock test helps you:
Improve accuracy by practising real NEET-level MCQs and reducing negative marking
Build speed and time management through a 3-hour exam-like simulation
Strengthen NCERT-based concepts with application-driven questions
Identify weak topics instantly for targeted same-day revision
Reduce exam anxiety by practising under real exam pressure
The biggest advantage? A same-day performance loop — Attempt → Analyse → Improve → Repeat — which is one of the most effective strategies followed by top NEET rankers in the final phase.
Consistently practising with this NEET 2026 mock test series ensures that your preparation converts into marks on the exam day, not just theoretical knowledge.
In the final days before NEET 2026, your score depends on how effectively you combine mock test practice, answer analysis, and same-day revision. A structured daily routine ensures faster improvement, better retention, and consistent score growth.
Follow this high-impact NEET daily plan:
Morning (Get Mock Question paper - 10 AM):
Attempt a full-length NEET mock test (180 questions) in strict exam conditions to build accuracy and time management.
Afternoon:
Analyse your mistakes in detail. Identify weak chapters, concepts, and question patterns that are causing errors.
Evening (After 5 PM):
Check the answer key and detailed solutions. Understand the correct approach and avoid repeating the same mistakes.
Night:
Revise important formulas, NCERT key points, Biology diagrams, and frequently mistaken topics for stronger retention.
This same-day improvement cycle (Test → Analyse → Revise) is one of the most effective NEET preparation 2026 strategies, helping you convert daily practice into real exam marks.
By following this routine consistently, you ensure continuous progress, better accuracy, and stronger exam confidence in the final phase.
By Day 3 of your NEET 2026 mock test practice, patterns in your performance have already started to appear. Skipping today’s test means missing a critical opportunity to fix recurring mistakes before the actual exam.
At this stage, it’s not just about practice—it’s about correction and consistency.
If you skip Day 3, you risk:
Repeating the same mistakes identified in Day 1 and Day 2
Carrying weak topics into the final exam without improvement
Inconsistent accuracy despite continuous revision
Poor time allocation across Physics, Chemistry, and Biology
Day 3 is where preparation starts turning into performance.
Regularly attempting full-length mock tests for NEET 2026 helps you:
Break error patterns and improve accuracy daily
Stabilise your score after initial fluctuations
Strengthen weak areas identified in earlier tests
Build consistency under real exam pressure
By now, the goal is simple: don’t just attempt—improve with every test.
Missing one day in this phase can break your momentum. Staying consistent with Day 3 ensures your preparation is moving in the right direction—towards a higher NEET 2026 score.
By Day 3, your focus should shift from just attempting to correct recurring errors. Avoiding these mistakes can directly improve your NEET score:
Attempting the mock test without a strict 3-hour time limit
Checking answers during the test and breaking exam flow
Skipping post-test analysis (the biggest mistake)
Ignoring weak topics identified in Day 1 and Day 2
Over-attempting doubtful questions, increasing negative marking
Fixing these mistakes by Day 3 can lead to a visible jump in accuracy and score stability.
Your goal in today’s mock test is not perfection—it is measurable improvement.
Even a +10 to +20 marks increase today can significantly impact your final NEET rank.
Focus on:
Accuracy over attempts in the 180-question paper
Reducing silly mistakes to minimise negative marking
Better time distribution across Physics, Chemistry, and Biology
Learning from mistakes identified in previous mock tests
By Day 3, your preparation should evolve into a performance system:
Attempt → Analyse → Correct → Improve
This is not just another mock test.
This is your score-building phase before NEET 2026.
This mock test is ideal for:
Students scoring below 500 and aiming for a quick score jump
Students stuck at the same score in Day 1–2
Aspirants struggling with accuracy and negative marking
Students focusing on NCERT-based revision through MCQs
Also check:
Frequently Asked Questions (FAQs)
You should attempt the mock test before 10 AM to simulate the real NEET exam environment and improve time management under pressure.
The answer key and detailed solutions will be released after 5 PM on the same day for immediate performance analysis.
Yes, this mock test follows the latest NCERT-based NEET 2026 pattern, including subject-wise weightage and exam-level difficulty.
Day 3 focuses more on improving accuracy and reducing repeated mistakes identified in earlier tests, helping you stabilise your score in the final preparation phase.
Yes, the answer key PDF with detailed solutions will be available for download after 5 PM.
With proper same-day analysis, students often see a 10–20 mark improvement due to reduced mistakes and better time management.
On Question asked by student community
Hello Dear Student,
No, NEET is not compulsory for Bachelor of Physiotherapy (BPT) admissions for the 2026–27 academic session.
You can check, find and access more information here:
https://news.careers360.com/physiotherapy-neet-admission-1yr-internship-bpt-dr-title-allied-health-sciences-paramedical-courses-revamp-syllabus-ncahp-mohfw
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-mds-2025-seat-allotment-result
Hope it helps!
Hello Dear Student,
With a NEET MDS 2026 score of 503 and an AIR of around 4600, getting a government seat in highly competitive branches such as Orthodontics, Conservative Dentistry & Endodontics, or Prosthodontics may be difficult, as these specialties usually close at much better ranks.
However, you still have
Hello Dear Student,
Haryana NEET MDS 2026 counselling registrations are expected to begin in July 2026. The official schedule will be released by the Department of Medical Education and Research (DMER) Haryana after the national MCC MDS counselling process commences.
You can check, find and access more information here:
https://medicine.careers360.com/articles/haryana-neet-mds-counselling
Hi!
Given below is the link to access the NEET 2026 Physics Question Paper:
https://medicine.careers360.com/articles/neet-physics-question-paper
You can check the link below, apply relevant filters, find suitable resources related to NEET that will boost your overall preparation:
https://www.careers360.com/download/neet-sample-papers-previous-years
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs