JSS University Mysore Allied Sciences 2026
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs
Important Chemistry Formulas for NEET 2026: Preparing for NEET Chemistry requires not only conceptual clarity but also quick recall of formulas. Which chemistry formulas are most important for NEET 2026, and how many questions are directly based on them every year? An analysis of NEET previous years’ question papers shows that a significant number of questions are directly or indirectly based on standard formulas from Physical Chemistry and key concepts from Organic and Inorganic Chemistry.
This Story also Contains

A 5-year analysis (2021–2025) reveals that several formula-based questions are consistently asked from chapters like Mole Concept, Thermodynamics, Chemical Equilibrium, Electrochemistry, Chemical Kinetics, and Solutions. Since NEET is largely based on NCERT, mastering these important chemistry formulas for NEET 2026 becomes essential to improve speed, accuracy, and overall performance in the NEET exam.
Chemical formulas are the backbone of NEET preparation 2026, especially in Physical Chemistry. It is the section of chemistry in which direct formula applications determine accuracy and speed.
Based on a previous year analysis of NEET (2021-2025), nearly half of the chemistry section requires formula recall. So, for the NEET aspirants, mastering the Chemistry formula means:
Quick problem-solving in a limited time
Higher accuracy in formula-based questions
Confidence in high-weightage topics like Mole Concept and Atomic Structure.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
According to the expert's analysis on 5-year PYQs (2021-2025), NEET Chemistry is not just about concepts. It is more about formulas that unlock marks quickly. Every year, there are direct applications of formulas from Physical Chemistry, Inorganic Chemistry’s Trends and the Basics of Organic Chemistry.
Chapter‑wise NEET Chemistry formulas first have Physical Chemistry formulas in NEET. The NCERT Chemistry formulas for the NEET exam include high-weightage chapters. Thermodynamics, Equilibrium, Electrochemistry, and Chemical Kinetics dominate the formula-based weightage for NEET 2026.
Some Important Chemistry Formulas For NEET 2026 Exam based on a previous year analysis of NEET (2021-2025) are given below -
Thermodynamics Highest Formula Weightage
First Law:
Enthalpy relation:
Gibbs Free Energy:
Relation with equilibrium constant:
Equilibrium constant, relation and buffer equation are given below:
Equilibrium constant:
Relation:
Buffer equation (Henderson–Hasselbalch):
Nernst Equation, conductivity and Gibbs relation from Electrochemistry:
Nernst Equation:
Gibbs relation:
Conductivity:
Number of moles:
Avogadro’s relation:
de Broglie wavelength:
Energy levels (Bohr model):
Important Formula of Chemical Kinetics:
Rate law:
Half-life for first order reaction:
Arrhenius equation:
Solution and Colloidal Properties, like osmotic pressure, are given below:
Osmotic pressure:
Depression in freezing point:
Elevation in boiling point:
NEET Chemistry Important Formulas PDF |
The Chemistry NEET section has about 10 - 12 formula-based questions each year. It is mainly from the Physical Chemistry portion. According to the NEET Chemistry Chapter-wise Weightage, these formula-based Chemistry questions are the direct application questions where the NCERT Chemistry formulas are tested for quick calculations.
The major Chemistry formula-based chapters are -
Thermodynamics & Equilibrium - 2–3 questions
Electrochemistry & Redox - 2–3 questions
Chemical Kinetics & Solutions - 2–3 questions
Atomic Structure & Mole Concept - 1–2 questions
The chemistry chapter with the maximum formula weightage in NEET is Thermodynamics and Chemical Kinetics. It is part of the Physical Chemistry section. These chapters have consistently contributed to 3-4 formula-based questions each year.
Example in NEET 2025, there were 4 questions from these chapters -
Question: If the rate constant of a reaction is
21.0 s
69.3 s
23.1 s
210 s
Solution: Given that
Initial concentration
Final concentration
Rate constant
Since units of
So,
put value
Now,
Hence, the correct answer is option (2).
Question: If the half-life
10 minutes
2 minutes
4 minutes
5 minutes
Solution: For first order reaction-
Also half-life for a first-order is given by-
Hence, the correct answer is option (1).
Question : The standard heat of formation, in
[Given : standard heat of formation of
+220.5
-128.5
-133.0
+133
Solution: Given that
Apply Hess’s Law, consider the following reaction:
Enthalpy change for this reaction is the heat of crystallisation:
Using Hess's law:
Rearranged to solve for
Hence, the correct answer is option 2) -128.5.
Question:
Which of the following diagrams gives an accurate representation of the above reaction?
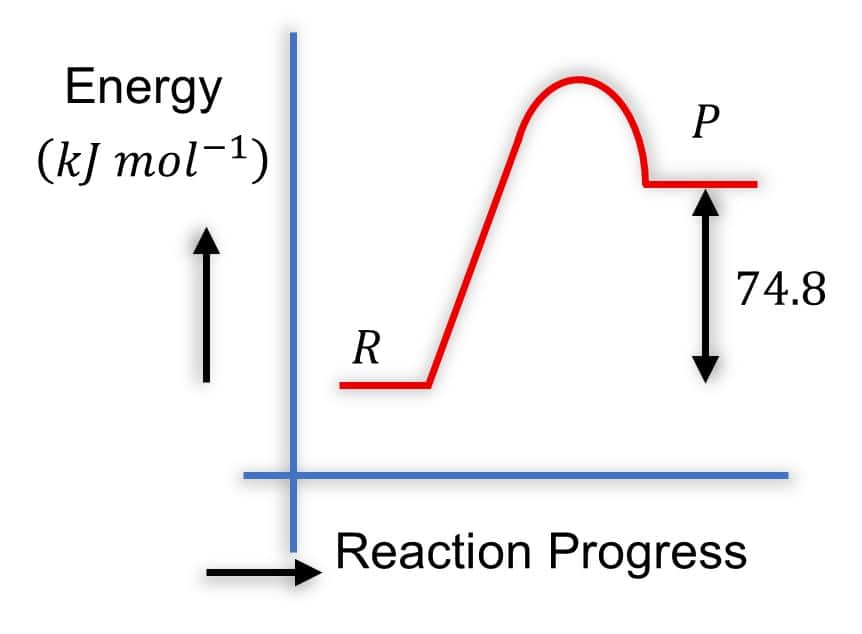
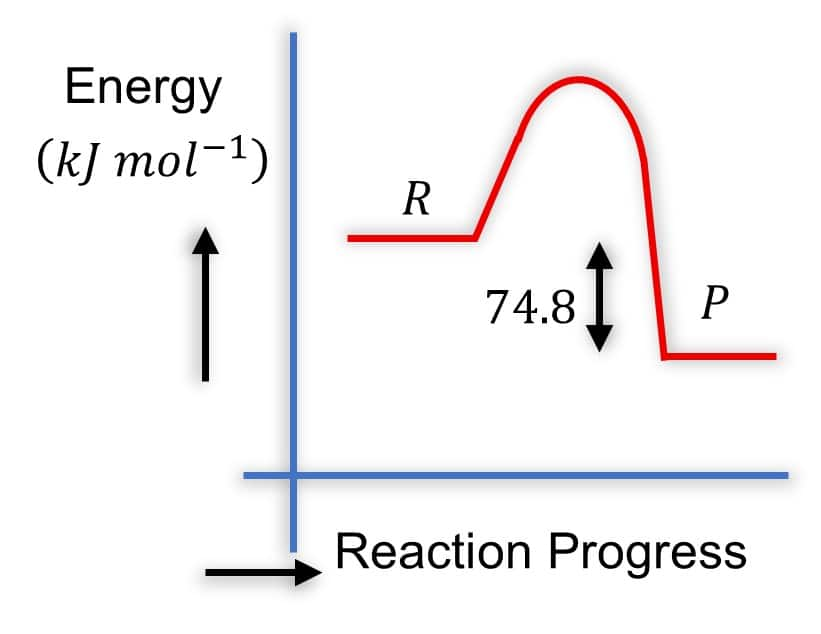
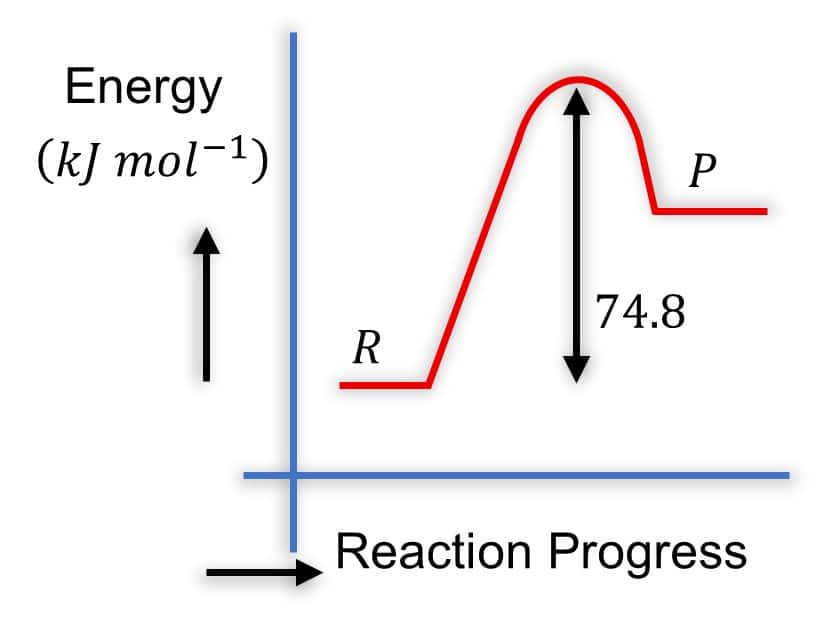
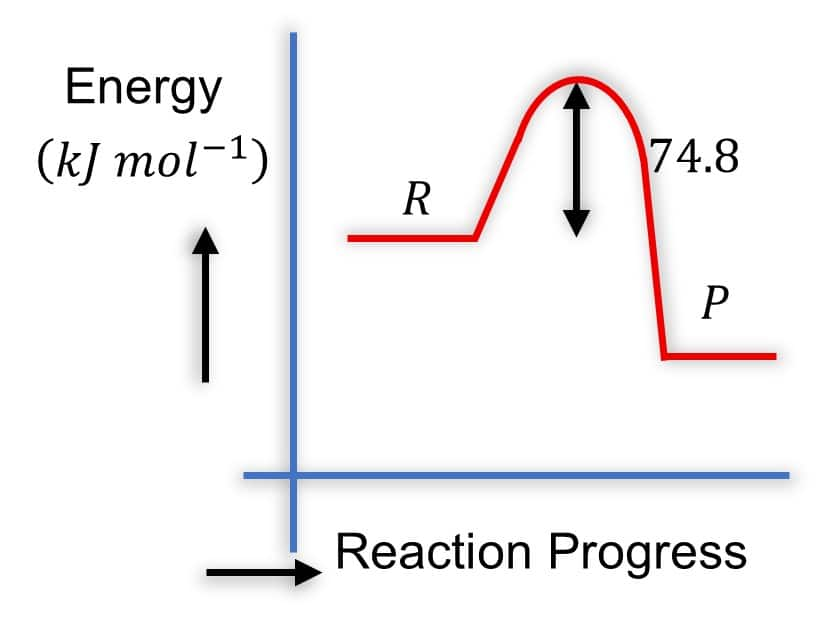
Solution: Diagram 2:
- R is higher than P
-
- The arrow from
- This is correct because it represents the energy difference, and
- It's common to show the magnitude of
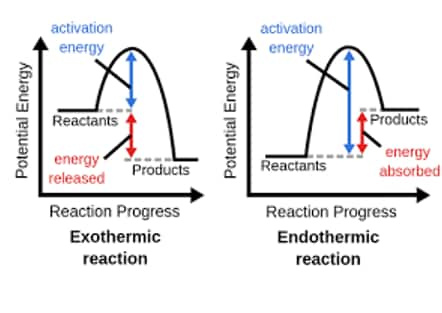
Hence, the correct answer is option (2).
Yes, the NCERT formulas are enough for NEET Chemistry, but only if you know how to apply them correctly. The reason is -
The NEET syllabus is NCERT-based. So almost all the formula-based chemistry questions in the physical chemistry section come straight from NCERT.
The NEET exam does not test the advanced derivations. It focuses on applying NCERT formulas to the numerical problems.
Also, year after year, these 10- 12 formula-based chemistry questions are solved directly using NCERT equations.
Frequently Asked Questions (FAQs)
Yes, but fewer compared to Physical Chemistry. Organic Chemistry uses general formulas and degree of unsaturation, while Inorganic Chemistry occasionally tests formulas like the solubility product.
You can download the NEET Chemistry 2026 Important Formulas PDF from this article. It includes chapter‑wise NCERT formulas for Thermodynamics, Equilibrium, Electrochemistry, Chemical Kinetics, Solutions, and Mole Concept.
On average, 10 - 12 formula‑based questions are asked in NEET Chemistry. Most of them come from Physical Chemistry chapters like Thermodynamics, Equilibrium, Electrochemistry, and Chemical Kinetics.
On Question asked by student community
Hello Dear Student,
Yes, qualifying for the NEET is mandatory if you are seeking admission to core medical courses like
MBBS
,
BDS
,
BAMS
,
BHMS
, and
BSc Nursing
across India.
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-eligibility-criteria
Hope it helps!
Hello Dear Student,
The NEET syllabus can seem overwhelming at first, but with a proper strategy and consistent effort, it becomes much more manageable. The key is to focus on the most important topics and practice regularly.
Effective NEET Preparation Tips
Prioritize NCERT Books: NCERT should be your primary study
Hello Dear Student,
No, NEET is not compulsory for Bachelor of Physiotherapy (BPT) admissions for the 2026–27 academic session.
You can check, find and access more information here:
https://news.careers360.com/physiotherapy-neet-admission-1yr-internship-bpt-dr-title-allied-health-sciences-paramedical-courses-revamp-syllabus-ncahp-mohfw
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-mds-2025-seat-allotment-result
Hope it helps!
Hello Dear Student,
You can check, find and access more information here:
https://medicine.careers360.com/articles/neet-2024-marks-vs-rank
https://medicine.careers360.com/articles/neet-2024-cut-off
Hope it helps!
Ranked among the top Dental Colleges for 7 consecutive years by India Today poll
Get Job Ready in Healthcare | Employability-Focused Programs
Allied & Healthcare programs | 20+ Partner Universities & Institutes | 98% placement record
Ranked as India’s #1 Not for profit pvt. University by India Today
Alied Health Sciences at SCSVMV | NAAC 'A' Grade | AICTE & UGC Aproved | 100% Placement Support | Merit-based Scholarships
NAAC A+ Accredited| Ranked #21 in University Category by NIRF | Applications open for multiple UG & PG Programs